QUESTION IMAGE
Question
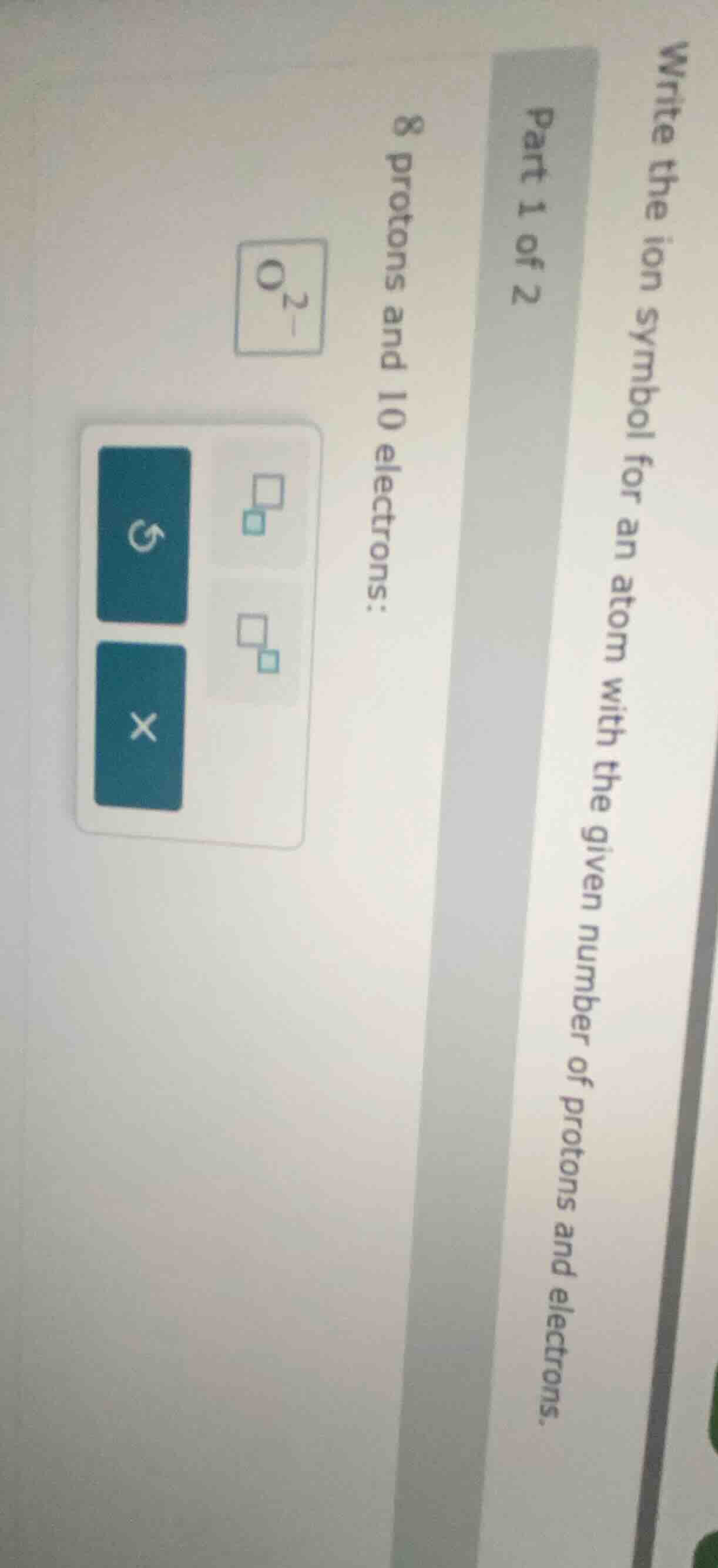
write the ion symbol for an atom with the given number of protons and electrons. part 1 of 2 8 protons and 10 electrons: o²⁻
Step1: Determine the element from protons
The number of protons is equal to the atomic number. Since there are 8 protons, the element is oxygen (O), as oxygen has an atomic number of 8.
Step2: Determine the charge from protons and electrons
The charge of an ion is calculated as (number of protons - number of electrons). Here, protons = 8 and electrons = 10. So charge = \(8 - 10 = -2\).
Step3: Write the ion symbol
The element is O, and the charge is -2, so the ion symbol is \(O^{2-}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(O^{2-}\)