QUESTION IMAGE
Question
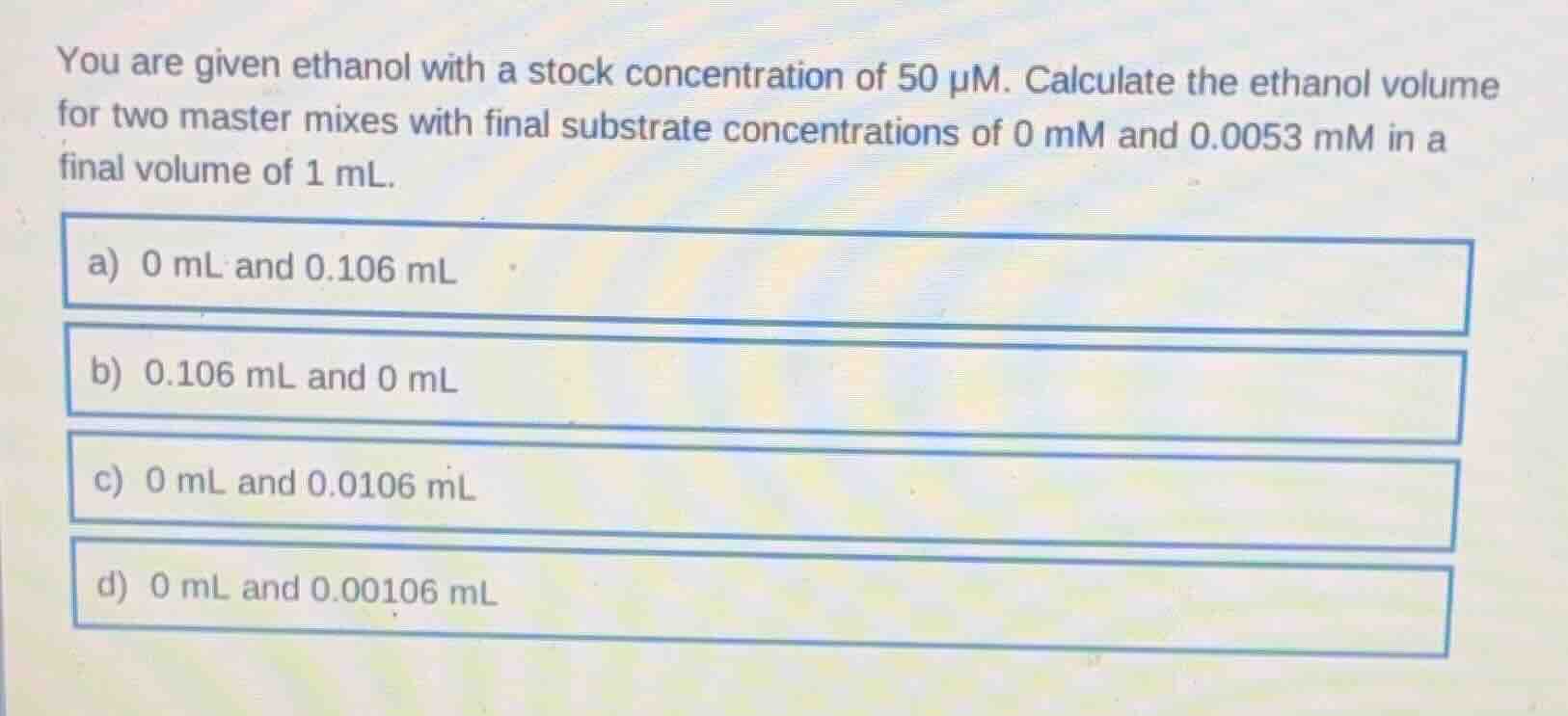
you are given ethanol with a stock concentration of 50 μm. calculate the ethanol volume for two master mixes with final substrate concentrations of 0 mm and 0.0053 mm in a final volume of 1 ml.
a) 0 ml and 0.106 ml
b) 0.106 ml and 0 ml
c) 0 ml and 0.0106 ml
d) 0 ml and 0.00106 ml
Step1: Convert units to match
Stock concentration: $50\ \mu\text{M} = 0.05\ \text{mM}$
Final volume $V_2 = 1\ \text{mL}$ for both mixes.
Step2: Calculate for 0 mM final conc.
A final concentration of 0 mM means no stock ethanol is added.
Volume of ethanol = $0\ \text{mL}$
Step3: Calculate for 0.0053 mM final conc.
Use dilution formula $C_1V_1 = C_2V_2$
Rearrange to solve for $V_1$: $V_1 = \frac{C_2V_2}{C_1}$
Substitute values: $V_1 = \frac{0.0053\ \text{mM} \times 1\ \text{mL}}{0.05\ \text{mM}} = 0.106\ \text{mL}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) 0 mL and 0.106 mL