QUESTION IMAGE
Question
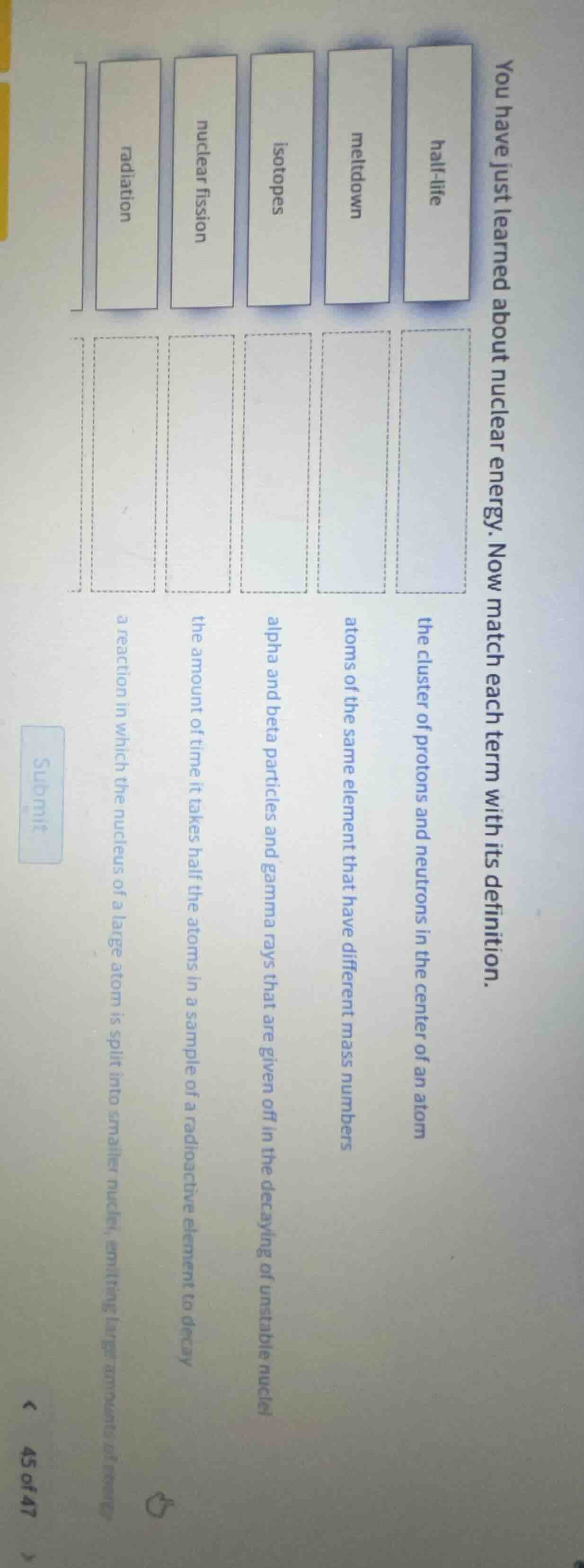
you have just learned about nuclear energy. now match each term with its definition.
half - life: the cluster of protons and neutrons in the center of an atom
meltdown: atoms of the same element that have different mass numbers
isotopes: alpha and beta particles and gamma rays that are given off in the decaying of unstable nuclei
nuclear fission: the amount of time it takes half the atoms in a sample of a radioactive element to decay
radiation: a reaction in which the nucleus of a large atom is split into smaller nuclei, emitting large amounts of energy
Matches are based on standard nuclear science definitions:
- Half-life describes radioactive decay timing.
- Isotopes are same-element atoms with different neutron counts (mass numbers).
- Radiation is the particles/rays released during nuclear decay.
- Nuclear fission is the splitting of heavy atomic nuclei for energy.
- The provided term "meltdown" (a reactor failure) does not align with the remaining definition, which refers to an atomic nucleus (not listed as a term).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- half-life → the amount of time it takes half the atoms in a sample of a radioactive element to decay
- isotopes → atoms of the same element that have different mass numbers
- radiation → alpha and beta particles and gamma rays that are given off in the decaying of unstable nuclei
- nuclear fission → a reaction in which the nucleus of a large atom is split into smaller nuclei, emitting large amounts of energy
- Note: The definition "the cluster of protons and neutrons in the center of an atom" corresponds to an atomic nucleus, which is not listed in the provided terms; the remaining term "meltdown" refers to a nuclear reactor failure event, which does not match any of the given definitions.