QUESTION IMAGE
Question
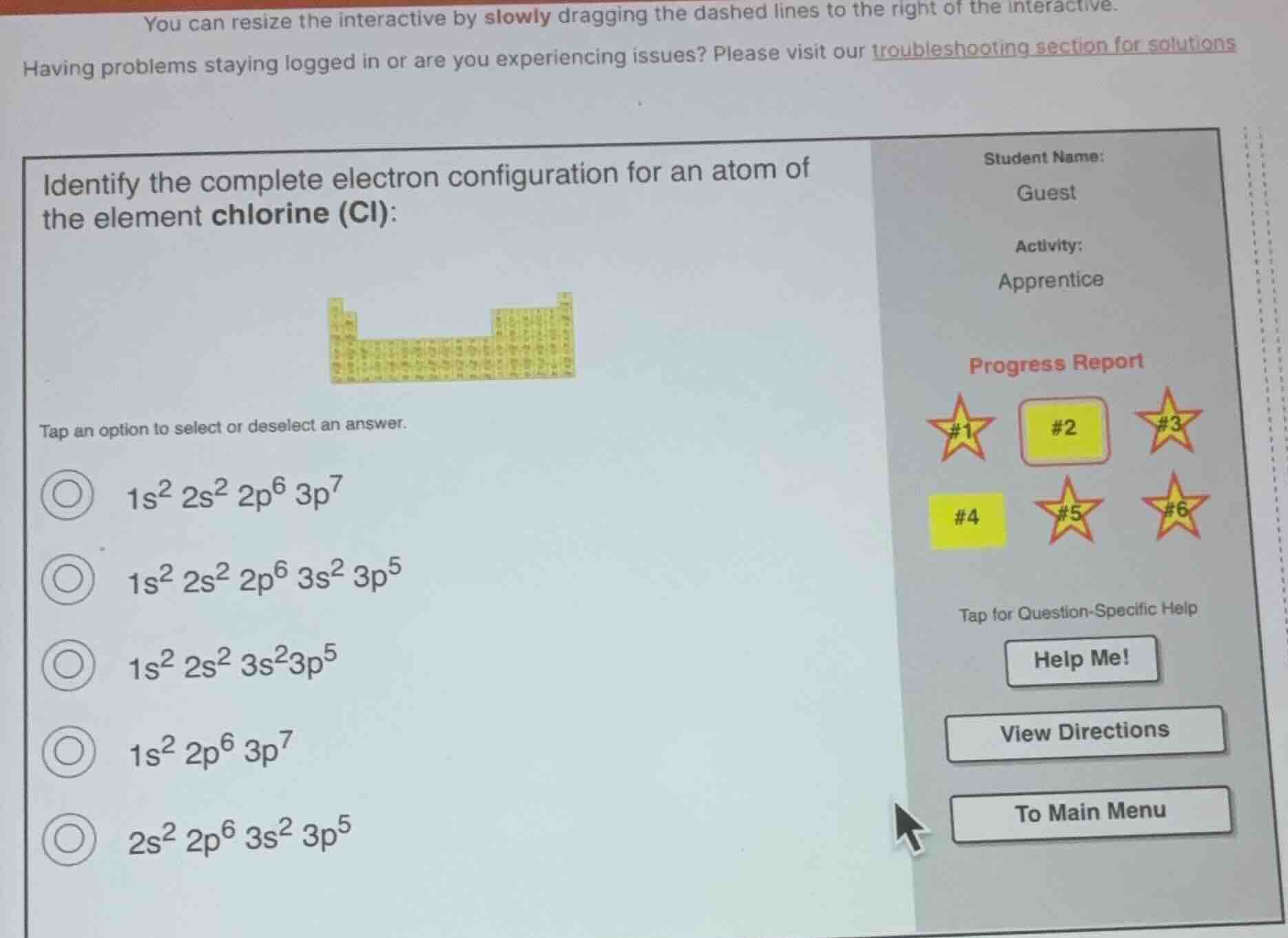
you can resize the interactive by slowly dragging the dashed lines to the right of the interactive.having problems staying logged in or are you experiencing issues? please visit our troubleshooting section for solutionsidentify the complete electron configuration for an atom ofthe element chlorine (cl):tap an option to select or deselect an answer.1s² 2s² 2p⁶ 3p⁷1s² 2s² 2p⁶ 3s² 3p⁵1s² 2s² 3s²3p⁵1s² 2p⁶ 3p⁷2s² 2p⁶ 3s² 3p⁵student name:guestactivity:apprenticeprogress report#1 #2 #3#4 #5 #6tap for question-specific helphelp me!view directionsto main menu
- First, find chlorine's atomic number: Chlorine (Cl) has an atomic number of 17, meaning a neutral chlorine atom has 17 electrons.
- Follow the aufbau principle to fill orbitals in order: 1s, 2s, 2p, 3s, 3p.
- Fill each orbital to its maximum capacity: 1s holds 2, 2s holds 2, 2p holds 6, 3s holds 2. The remaining 5 electrons go to 3p.
- Sum the exponents: $2+2+6+2+5=17$, which matches the total number of electrons. Eliminate options that skip orbitals, have incorrect orbital filling order, or the wrong total number of electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1s^2 2s^2 2p^6 3s^2 3p^5$ (the second option)