QUESTION IMAGE
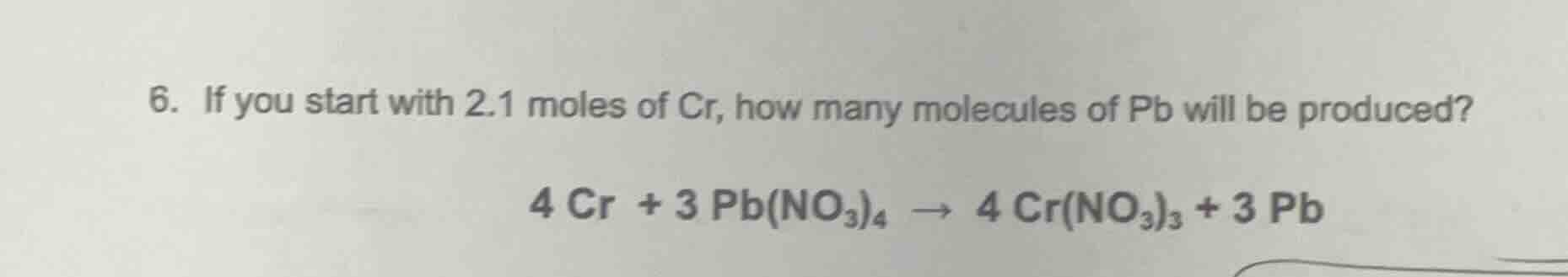
Question
- if you start with 2.1 moles of cr, how many molecules of pb will be produced?
$4\\ cr + 3\\ pb(no_3)_4 \
ightarrow 4\\ cr(no_3)_3 + 3\\ pb$
Step1: Find mole ratio of Cr to Pb
From the balanced equation, the mole ratio is $\frac{3\ \text{mol Pb}}{4\ \text{mol Cr}}$.
Step2: Calculate moles of Pb
Multiply starting moles of Cr by the ratio:
$2.1\ \text{mol Cr} \times \frac{3\ \text{mol Pb}}{4\ \text{mol Cr}} = 1.575\ \text{mol Pb}$
Step3: Convert moles to molecules
Use Avogadro's number $6.022 \times 10^{23}\ \text{molecules/mol}$:
$1.575\ \text{mol Pb} \times 6.022 \times 10^{23}\ \frac{\text{molecules}}{\text{mol}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$9.48 \times 10^{23}$ molecules of Pb (rounded to three significant figures)