QUESTION IMAGE
Question
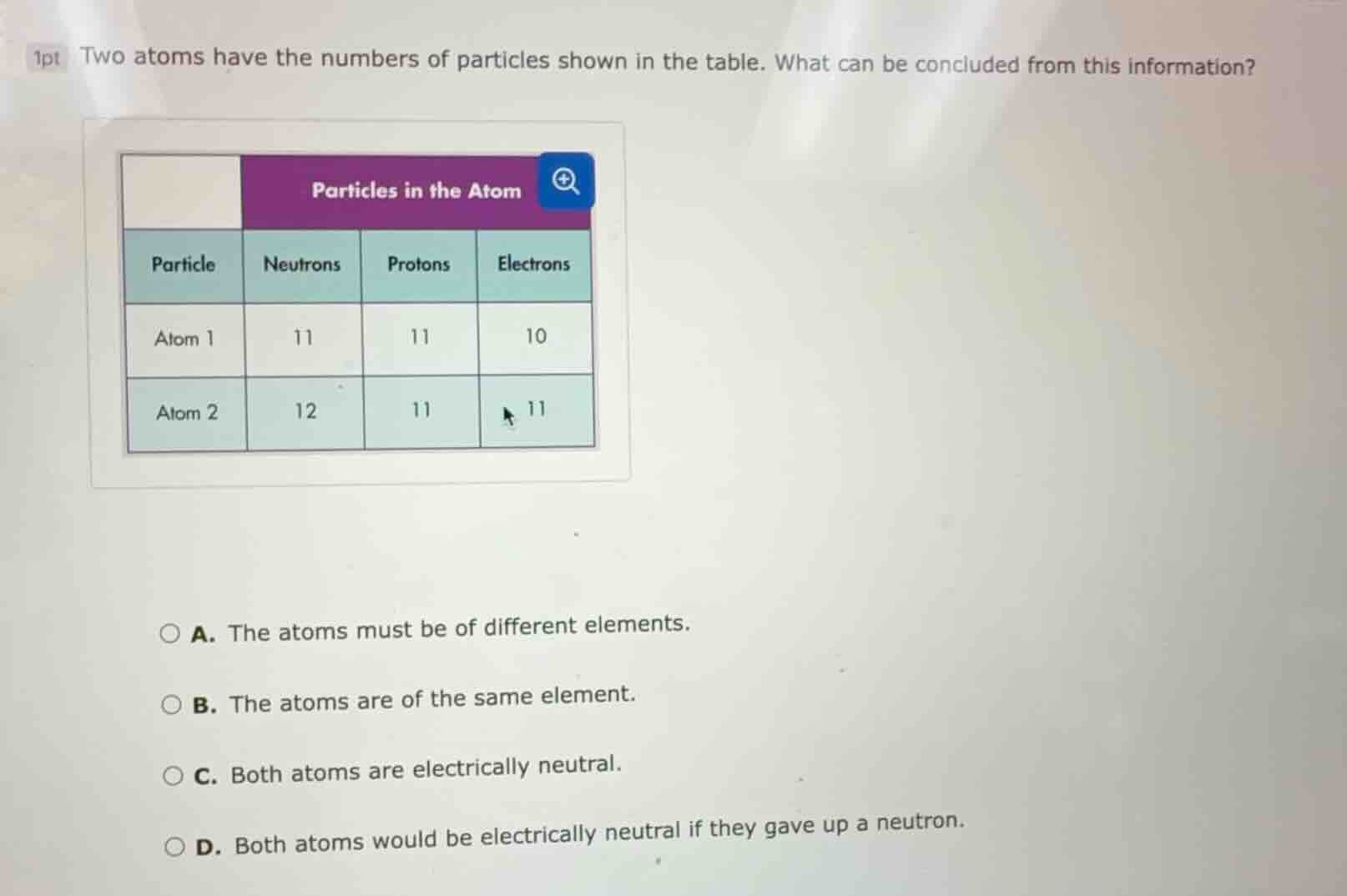
1pt two atoms have the numbers of particles shown in the table. what can be concluded from this information?
particles in the atom
particle neutrons protons electrons
atom 1 11 11 10
atom 2 12 11 11
a. the atoms must be of different elements.
b. the atoms are of the same element.
c. both atoms are electrically neutral.
d. both atoms would be electrically neutral if they gave up a neutron.
Brief Explanations
- The identity of an element is determined by the number of protons in an atom's nucleus. Both atoms have 11 protons, so they are the same element (sodium).
- An atom is electrically neutral if the number of protons (positive charge) equals the number of electrons (negative charge). Atom 1 has 11 protons and 10 electrons, so it has a +1 charge (not neutral). Atom 2 has equal protons and electrons, so it is neutral.
- Neutrons have no electrical charge, so changing the number of neutrons does not affect an atom's electrical neutrality.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The atoms are of the same element.