QUESTION IMAGE
Question
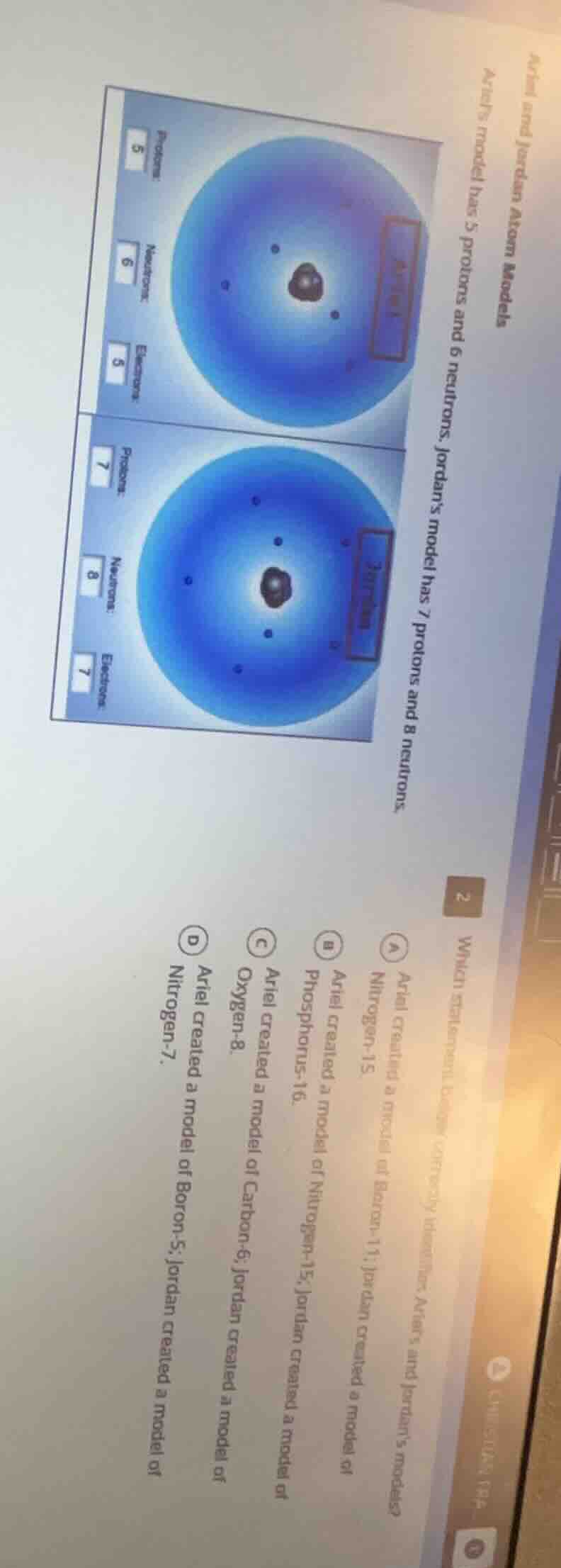
ariel and jordan atom models
ariel’s model has 5 protons and 6 neutrons. jordan’s model has 7 protons and 8 neutrons.
2
which statement correctly identifies ariel’s and jordan’s models?
a ariel created a model of boron-11; jordan created a model of nitrogen-15.
b ariel created a model of nitrogen-15; jordan created a model of phosphorus-16.
c ariel created a model of carbon-6; jordan created a model of oxygen-8.
d ariel created a model of boron-5; jordan created a model of nitrogen-7.
To determine the correct statement, we analyze the atomic models:
- Ariel’s model: 5 protons (atomic number = 5, Boron) and 6 neutrons. Mass number = protons + neutrons = \( 5 + 6 = 11 \), so Boron - 11.
- Jordan’s model: 7 protons (atomic number = 7, Nitrogen) and 8 neutrons. Mass number = \( 7 + 8 = 15 \), so Nitrogen - 15.
Now, check the options:
- Option A: Ariel (Boron - 11) and Jordan (Nitrogen - 15) – matches our analysis.
- Option B: Jordan’s model is Nitrogen - 15, not Phosphorus - 16 (Phosphorus has atomic number 15, not 7). Eliminate.
- Option C: Ariel’s protons (5) do not match Carbon (atomic number 6) or Oxygen (atomic number 8). Eliminate.
- Option D: Jordan’s model is Nitrogen - 15, not Nitrogen - 7 (mass number \( 7 + 8 = 15 \), not 7). Eliminate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Ariel created a model of Boron - 11; Jordan created a model of Nitrogen - 15.