QUESTION IMAGE
Question
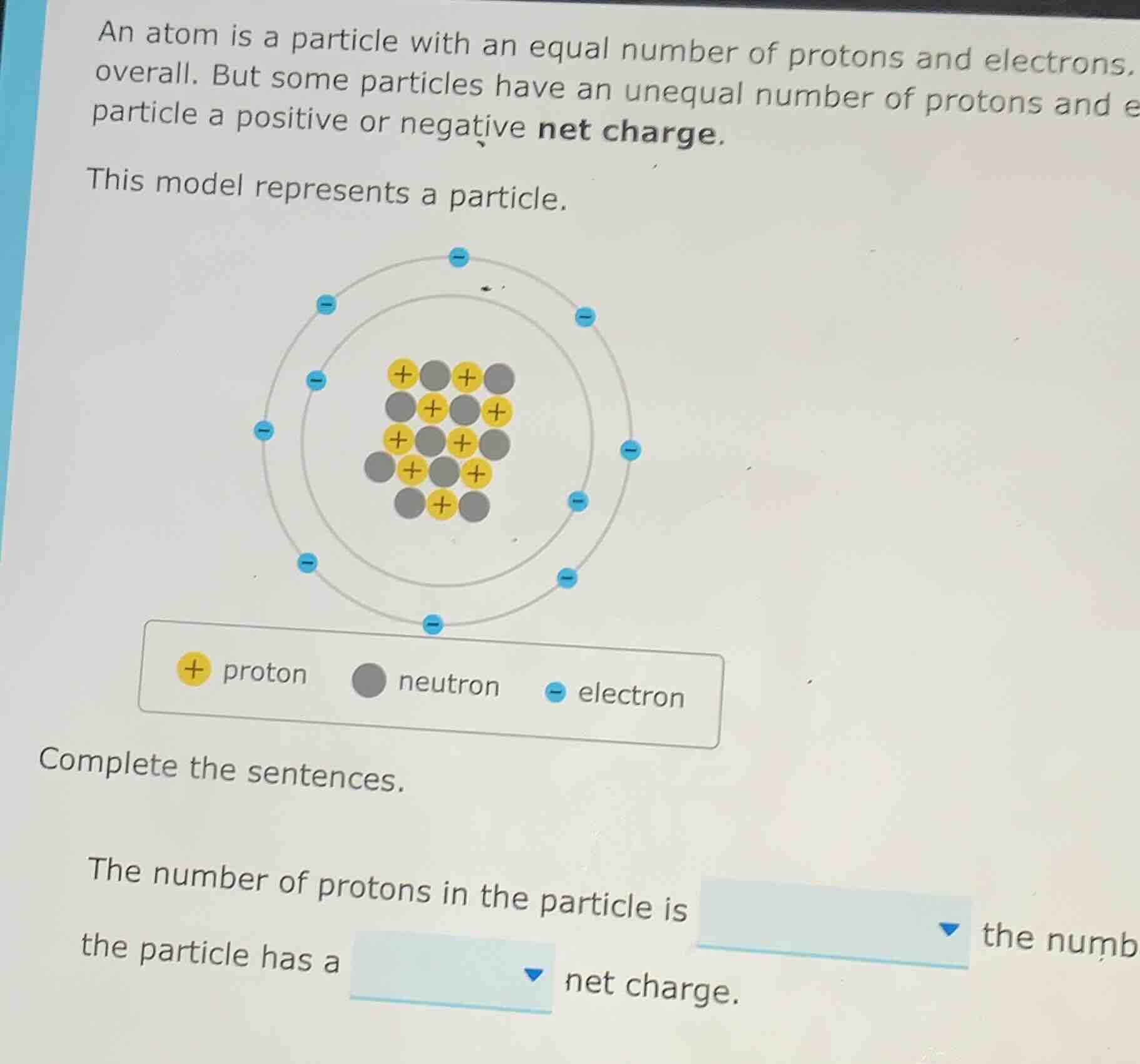
an atom is a particle with an equal number of protons and electrons, overall. but some particles have an unequal number of protons and e particle a positive or negative net charge. this model represents a particle. complete the sentences. the number of protons in the particle is the numb the particle has a net charge.
Step1: Count protons in nucleus
Count the yellow (+) particles: $1+1+1+1+1+1+1+1+1+1+1 = 11$
Step2: Count electrons in shells
Count the blue (-) particles: $1+1+1+1+1+1+1+1+1+1+1+1 = 12$
Step3: Compare protons and electrons
Protons = 11, Electrons = 12. $11 < 12$
Step4: Calculate net charge
Net charge = (Number of protons) - (Number of electrons) = $11 - 12 = -1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number of protons in the particle is less than the number of electrons, so the particle has a negative net charge.