QUESTION IMAGE
Question
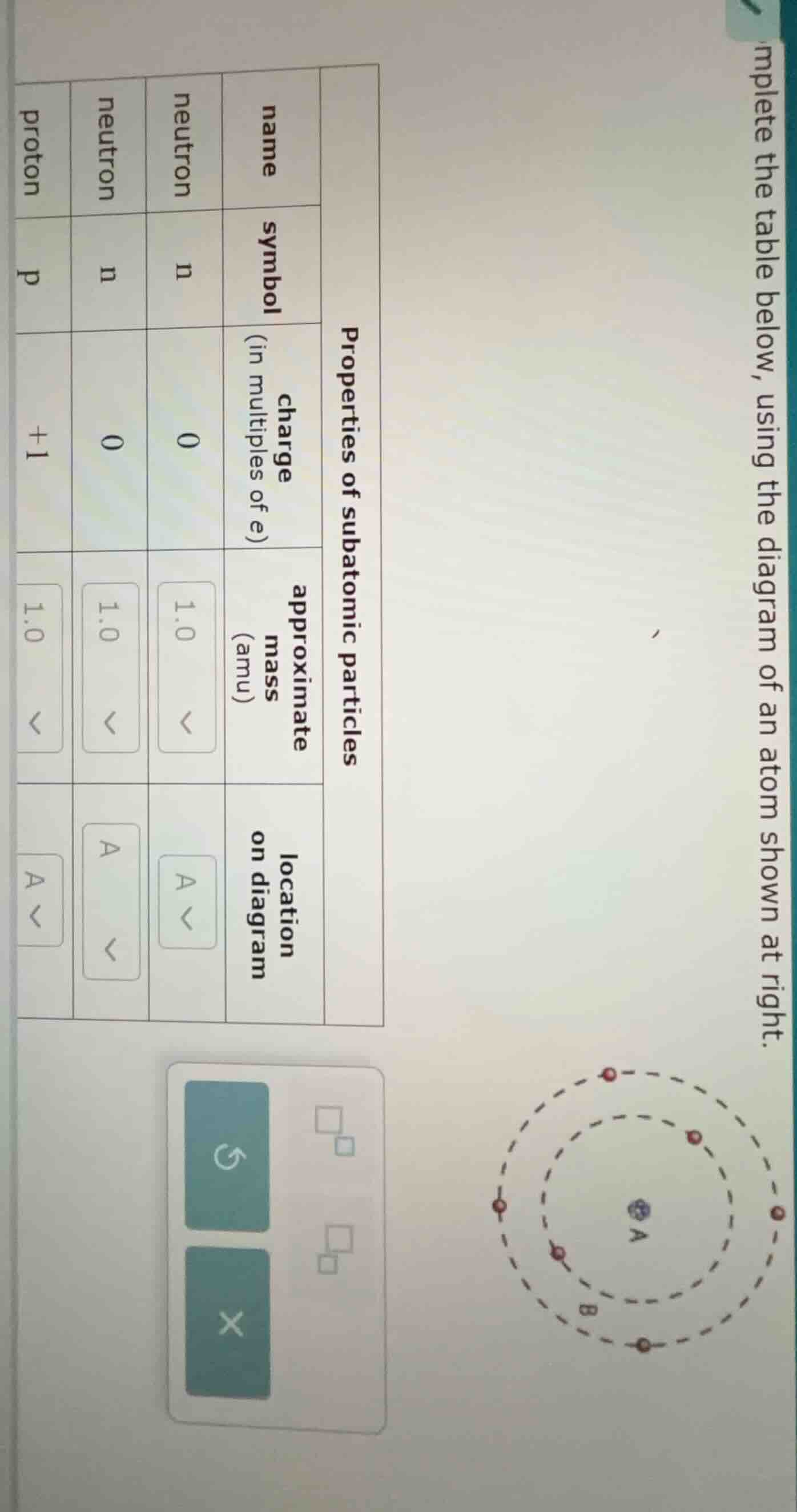
complete the table below, using the diagram of an atom shown at right.
properties of subatomic particles
| name | symbol | charge (in multiples of e) | approximate mass (amu) | location on diagram | |
|---|---|---|---|---|---|
| neutron | n | 0 | 1.0 | ||
| proton | p | +1 | 1.0 |
Step1: Identify the first missing particle name
Looking at the table, the first row under "name" has a missing entry. The symbol is "n" and charge is 0, mass ~1 amu. This is a neutron? Wait, no, the first column after name: the first particle with symbol "n" – wait, the table has columns: name, symbol, charge, mass, location. The first row (after header) has name missing, symbol "n", charge 0, mass ~1 amu. So that's a neutron? Wait, no, the second row is neutron (name: neutron, symbol n). Wait, the first row: name is missing, symbol n, charge 0, mass ~1 amu. Wait, maybe it's a typo or maybe the first particle is neutron? Wait, no, protons are p, neutrons n. Wait, the table has:
First column (name): first cell empty, second neutron, third neutron, fourth proton.
Wait, no, let's re-express the table:
| name | symbol | charge (multiples of e) | approximate mass (amu) | location on diagram | |
|---|---|---|---|---|---|
| Row 2 | neutron | n | 0 | ~1 | |
| Row 3 | proton | p | +1 | ~1 |
Wait, maybe the first particle is also a neutron? Wait, no, maybe the first row is a neutron (same as row 2). Wait, the diagram: the center (A) is the nucleus, which contains protons and neutrons. The electrons are in the orbits (B and outer).
So for the location:
- Neutrons and protons are in the nucleus (location A).
- Electrons are in the electron cloud/orbits (location B or outer).
Now, let's fill the table:
- First row (name): neutron (since symbol n, charge 0, mass ~1 amu, same as row 2)
- Charge for proton is +1 (already filled)
- Approximate mass: proton and neutron are both ~1 amu (proton is ~1.007, neutron ~1.008, but approximate ~1)
- Location:
- Neutrons (row 1, 2) and proton (row 3) are in the nucleus (location A)
- Electrons (not in table, but if there were, they'd be in B/outer)
Wait, the table has three particles: two neutrons and one proton? Wait, maybe the first particle is a neutron (name: neutron), symbol n, charge 0, mass ~1, location A (nucleus).
Row 1: name = neutron, symbol n, charge 0, mass ~1, location A.
Row 2: name = neutron, symbol n, charge 0, mass ~1, location A.
Row 3: name = proton, symbol p, charge +1, mass ~1, location A.
Wait, but the diagram: A is the nucleus (protons and neutrons), B is the inner electron shell, outer is outer shell.
So for the location column:
- Neutrons (row 1, 2) and proton (row 3) are in A (nucleus).
Now, let's check the approximate mass: protons and neutrons both have approximate mass ~1 amu (proton: ~1.007, neutron: ~1.008, so ~1 for approximation).
Charge: neutrons 0, proton +1.
So filling the table:
First row (name): neutron
Charge for first row: 0 (already filled)
Approximate mass: ~1 (already filled with <, maybe indicating ~1)
Location: A (nucleus)
Second row (neutron): location A
Third row (proton): location A
Wait, maybe the first particle is a neutron, same as the second. So the missing name in row 1 is "neutron".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The completed table (relevant parts):
| name | symbol | charge (multiples of e) | approximate mass (amu) | location on diagram |
|---|---|---|---|---|
| neutron | n | 0 | ~1 | A |
| proton | p | +1 | ~1 | A |
(Note: The first row's name is "neutron", location is "A" (nucleus), same as neutron and proton since they are in the nucleus. Electrons would be in the orbits (B or outer), but they are not in this table.)