QUESTION IMAGE
Question
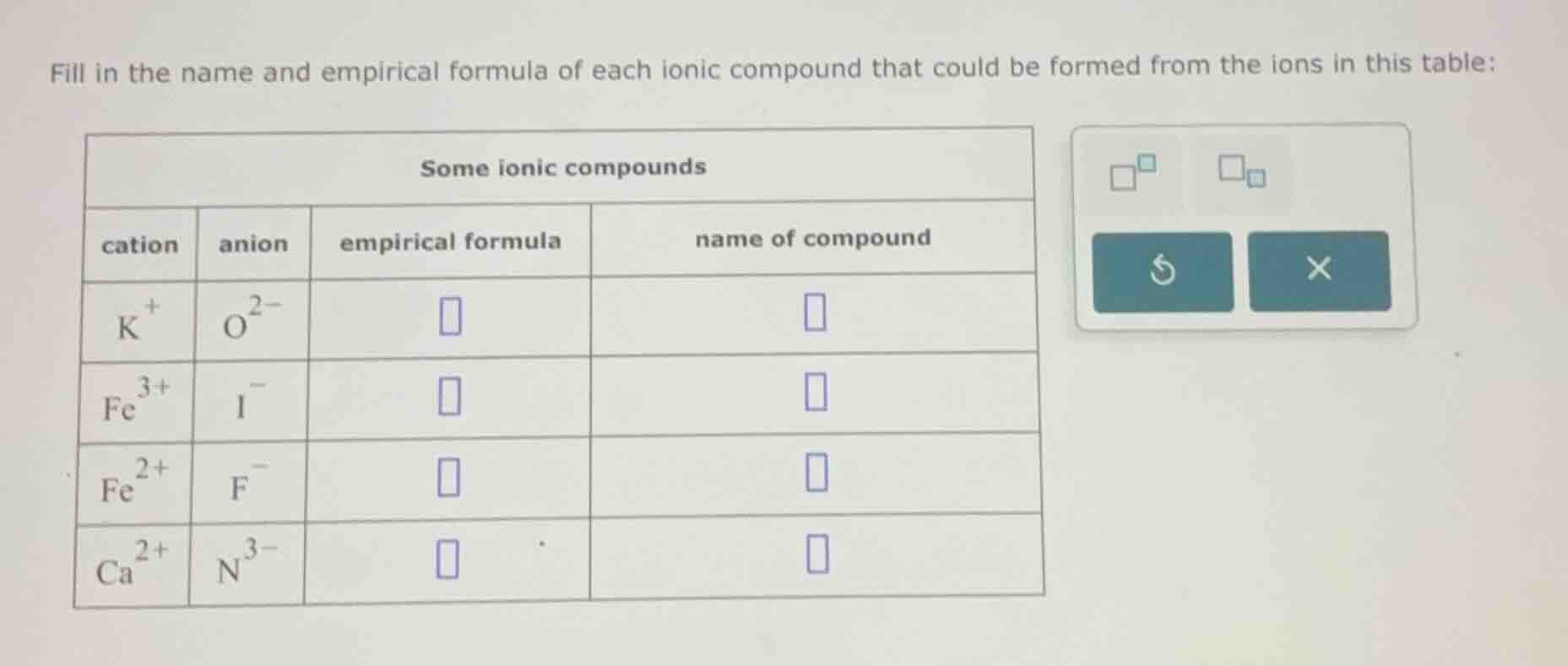
fill in the name and empirical formula of each ionic compound that could be formed from the ions in this table:
some ionic compounds
cation | anion | empirical formula | name of compound
$\text{k}^+$ | $\text{o}^{2-}$ | |
$\text{fe}^{3+}$ | $\text{i}^-$ | |
$\text{fe}^{2+}$ | $\text{f}^-$ | |
$\text{ca}^{2+}$ | $\text{n}^{3-}$ | |
Step1: Balance charges for K⁺ & O²⁻
To balance +1 and -2 charges, use 2 K⁺ and 1 O²⁻. Formula: $\text{K}_2\text{O}$; Name: Potassium oxide (K is group 1, fixed charge).
Step2: Balance charges for Fe³⁺ & I⁻
To balance +3 and -1 charges, use 1 Fe³⁺ and 3 I⁻. Formula: $\text{FeI}_3$; Name: Iron(III) iodide (Fe has variable charge, specify +3).
Step3: Balance charges for Fe²⁺ & F⁻
To balance +2 and -1 charges, use 1 Fe²⁺ and 2 F⁻. Formula: $\text{FeF}_2$; Name: Iron(II) fluoride (Fe has variable charge, specify +2).
Step4: Balance charges for Ca²⁺ & N³⁻
To balance +2 and -3 charges, use 3 Ca²⁺ and 2 N³⁻. Formula: $\text{Ca}_3\text{N}_2$; Name: Calcium nitride (Ca is group 2, fixed charge).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| cation | anion | empirical formula | name of compound |
|---|---|---|---|
| $\text{Fe}^{3+}$ | $\text{I}^-$ | $\text{FeI}_3$ | Iron(III) iodide |
| $\text{Fe}^{2+}$ | $\text{F}^-$ | $\text{FeF}_2$ | Iron(II) fluoride |
| $\text{Ca}^{2+}$ | $\text{N}^{3-}$ | $\text{Ca}_3\text{N}_2$ | Calcium nitride |