QUESTION IMAGE
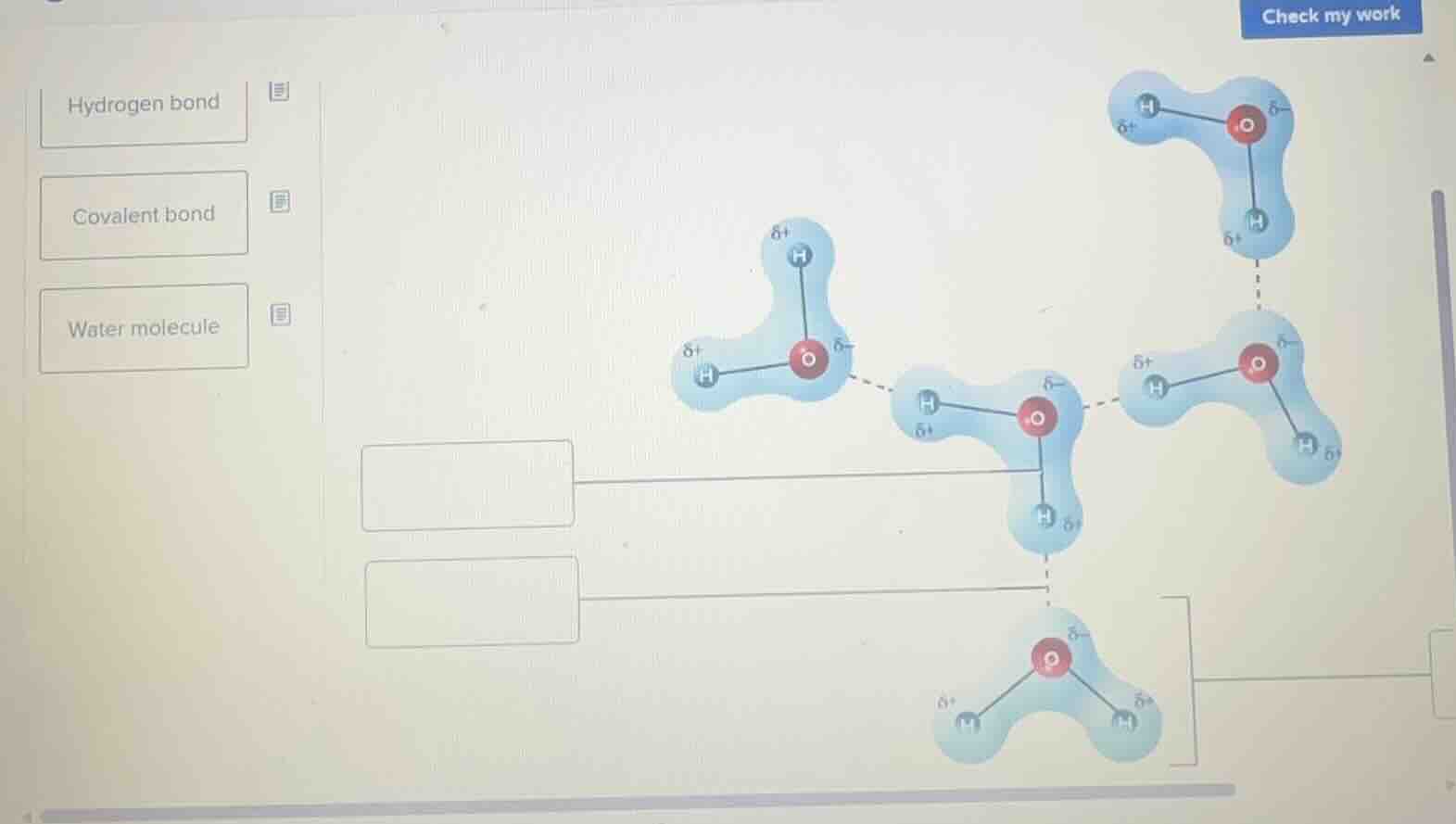
Question
hydrogen bond
covalent bond
water molecule
check my work
Brief Explanations
- The lines connecting the oxygen and hydrogen atoms within a single blue outlined structure represent covalent bonds, as these are the intramolecular bonds that hold the atoms of a single water molecule together.
- The dashed lines connecting the partially positive hydrogen of one water molecule to the partially negative oxygen of a neighboring water molecule represent hydrogen bonds, as these are the intermolecular attractions between separate water molecules.
- Each single blue outlined structure containing one oxygen and two hydrogen atoms is a water molecule.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Top empty box (connecting to the solid lines between O and H in a single structure): Covalent bond
- Bottom empty box (connecting to the dashed line between two structures): Hydrogen bond
- Right empty box (connecting to one full blue O-H structure): Water molecule