QUESTION IMAGE
Question
1.1 measurements
what information does every measurement provide?
what are si units?
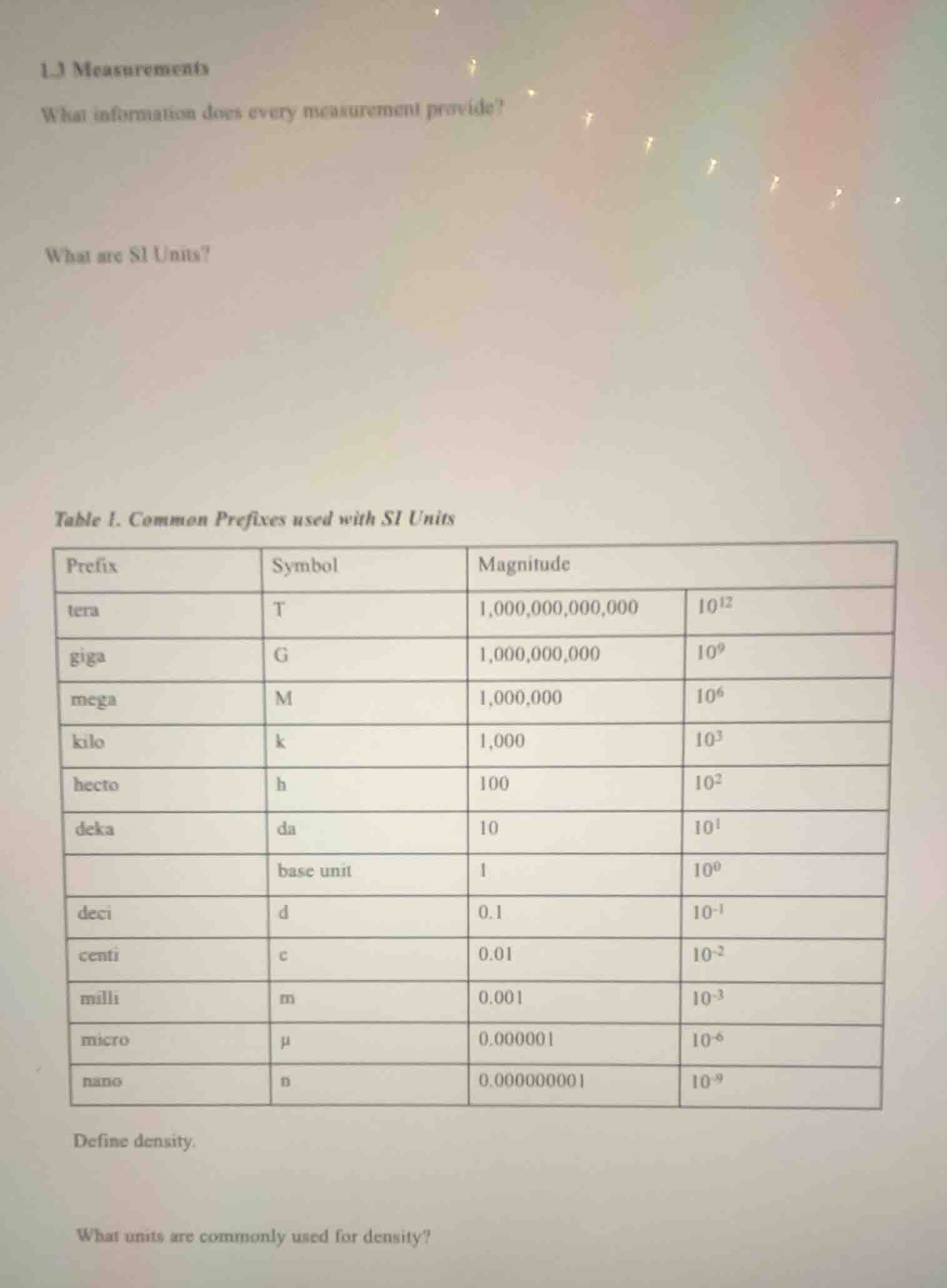
table 1. common prefixes used with si units
prefix | symbol | magnitude
--- | --- | --- | ---
tera | t | 1,000,000,000,000 | $10^{12}$
giga | g | 1,000,000,000 | $10^9$
mega | m | 1,000,000 | $10^6$
kilo | k | 1,000 | $10^3$
hecto | h | 100 | $10^2$
deka | da | 10 | $10^1$
| base unit | 1 | $10^0$ |
deci | d | 0.1 | $10^{-1}$
centi | c | 0.01 | $10^{-2}$
milli | m | 0.001 | $10^{-3}$
micro | $\mu$ | 0.000001 | $10^{-6}$
nano | n | 0.000000001 | $10^{-9}$
define density.
what units are commonly used for density?
- For the first question, every measurement must communicate two key details: a numerical value (how much) and a unit (the standard of comparison).
- For the second question, SI Units are the globally accepted standardized measurement system, defined by the International Bureau of Weights and Measures, with 7 base units that all other units are derived from.
- For the third question, density is a physical property that quantifies the concentration of mass in a given volume.
- For the fourth question, common density units are based on mass and volume combinations, with metric/SI and common non-SI options.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Every measurement provides a numerical magnitude (a quantitative value) and a standard unit (to define the scale of the measurement).
- SI Units (International System of Units) are the global standard for scientific and general measurements, consisting of 7 base units (e.g., meter for length, kilogram for mass) and derived units, ensuring consistency across disciplines and regions.
- Density is the amount of mass per unit volume of a substance, mathematically expressed as $
ho = \frac{m}{V}$ where $
ho$ = density, $m$ = mass, $V$ = volume.
- Commonly used units for density include:
- SI unit: $\frac{\text{kg}}{\text{m}^3}$ (kilograms per cubic meter)
- Common metric unit: $\frac{\text{g}}{\text{cm}^3}$ (grams per cubic centimeter)
- Common non-metric unit: $\frac{\text{lb}}{\text{ft}^3}$ (pounds per cubic foot)