QUESTION IMAGE
Question
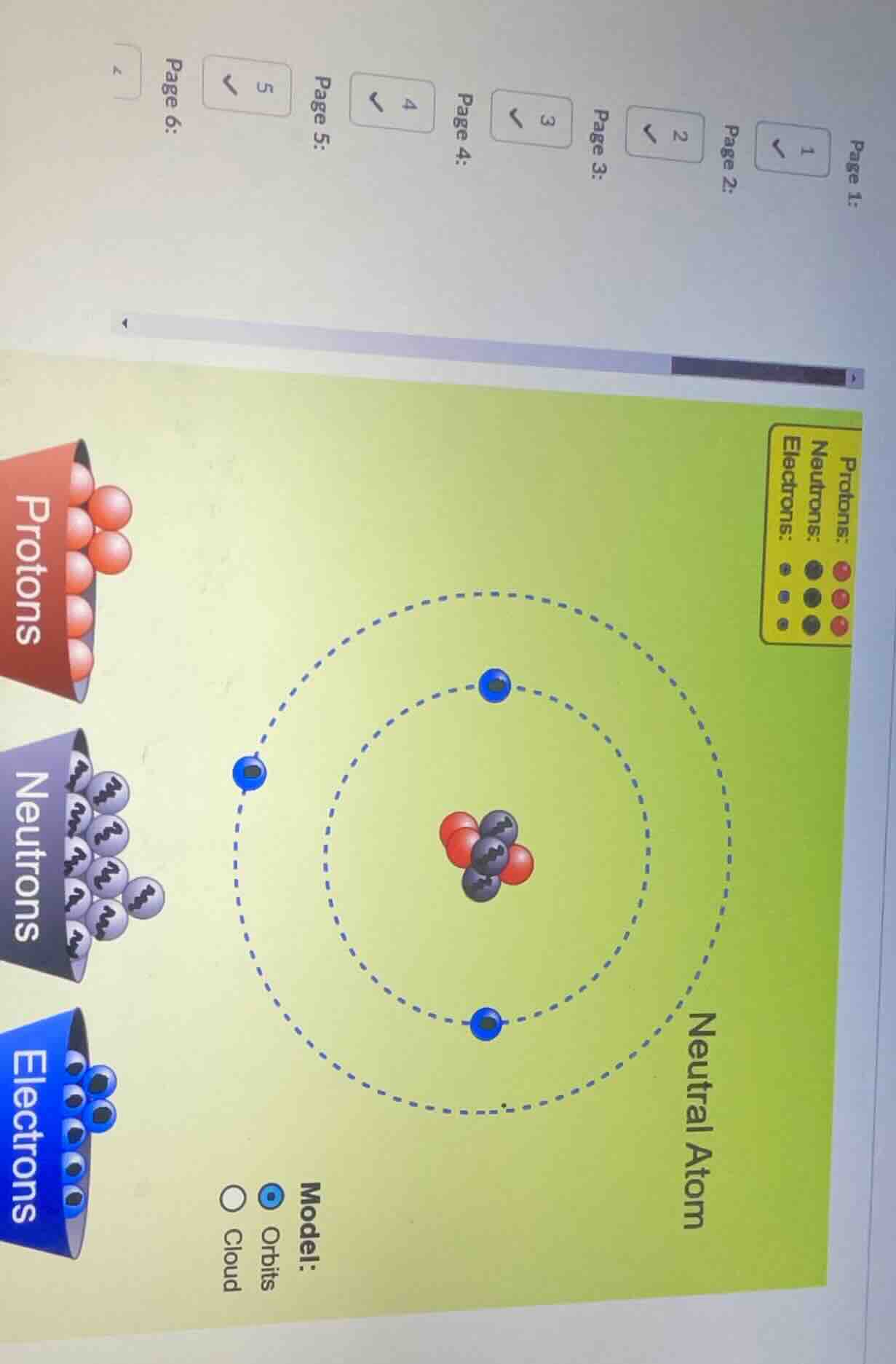
protons neutrons electrons neutral atom model: orbits cloud
Since the problem (related to atomic structure with protons, neutrons, electrons) is not fully stated (e.g., what to calculate or analyze), we can't proceed. But if we assume a common question like "Find the atomic number (Z) and mass number (A) of this atom", here's the solution:
Step1: Determine number of protons (Z)
From the diagram, count protons (red spheres in nucleus). There are 3 protons. So atomic number \( Z = 3 \).
Step2: Determine number of neutrons (N)
Count neutrons (gray spheres in nucleus). There are 3 neutrons.
Step3: Calculate mass number (A)
Mass number \( A = Z + N \). Substitute \( Z = 3 \), \( N = 3 \): \( A = 3 + 3 = 6 \).
Step4: Check electrons (for neutral atom)
Electrons (blue spheres in orbits) count: 3. For neutral atom, electrons = protons, which matches (\( 3 = 3 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Atomic number (\( Z \)) = 3, Mass number (\( A \)) = 6. The atom is Lithium - 6 (since \( Z = 3 \) is Lithium).
If the actual question was different (e.g., identify the atom, check charge, etc.), the approach would adjust. Since the original problem's question is missing, this is a common atomic structure - related solution based on the diagram.