QUESTION IMAGE
Question
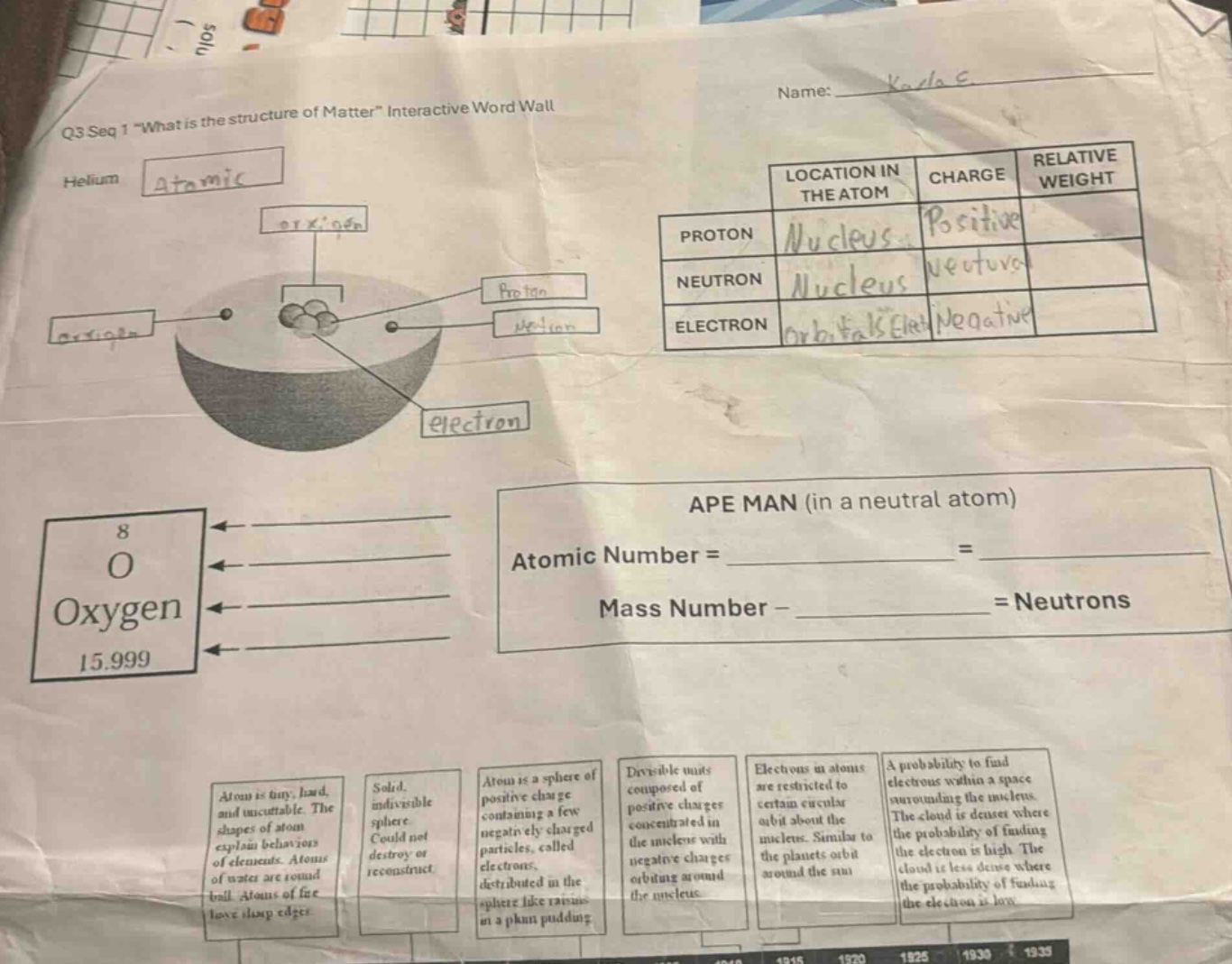
q3 seq 1 \what is the structure of matter\ interactive word wall
name: karla e.
helium
atomic
oxygen
proton
neutron
oxygen
electron
location in the atom
charge
relative weight
proton nucleus positive
neutron nucleus neutral
electron orbitals/shells negative
8
o
oxygen
15.999
ape man (in a neutral atom)
atomic number =
=
mass number -
= neutrons
atom is tiny, hard, and uncuttable. the shapes of atom explain behaviors of elements. atoms of water are round ball. atoms of fire have sharp edges.
solid, indivisible sphere
could not destroy or reconstruct
atom is a sphere of positive charge containing a few negatively charged particles, called electrons, distributed in the sphere like raisins in a plum pudding
divisible units composed of positive charges concentrated in the nucleus with negative charges orbiting around the nucleus.
electrons in atoms are restricted to certain circular orbit about the nucleus. similar to the planets orbit around the sun
a probability to find electrons within a space surrounding the nucleus. the cloud is denser where the probability of finding the electron is high. the cloud is less dense where the probability of finding the electron is low
1870 1875 1930 1935
For the "APE MAN (in a neutral atom)" section:
Atomic Number:
In a neutral atom, the atomic number is equal to the number of protons, and also equal to the number of electrons (since the atom is neutral, positive and negative charges balance). For an oxygen atom (as shown with the symbol \( \ce{O} \) and atomic number 8), the atomic number is defined as the number of protons in the nucleus. So, Atomic Number = Number of Protons = Number of Electrons (in a neutral atom).
Neutrons:
The formula to find the number of neutrons in an atom is: Mass Number - Atomic Number = Neutrons. The mass number is the sum of protons and neutrons in the nucleus. For oxygen, the mass number can be approximated from the atomic mass (15.999, which we can round to 16 for simplicity). The atomic number of oxygen is 8. So, Neutrons = Mass Number - Atomic Number.
Filling in the blanks:
- Atomic Number = Number of Protons = Number of Electrons (in a neutral atom). For oxygen, the atomic number is 8, so Atomic Number = \( \boldsymbol{8} \) (number of protons) = \( \boldsymbol{8} \) (number of electrons, since it's neutral).
- Mass Number – Atomic Number = Neutrons. Using oxygen's mass number (≈16) and atomic number (8), Neutrons = \( 16 - 8 = 8 \) (for oxygen, this is a simplified example; the actual number of neutrons in oxygen can vary slightly in isotopes, but the formula holds).
For the table (location, charge, relative weight of subatomic particles):
| Particle | Location in the Atom | Charge | Relative Weight |
|---|---|---|---|
| Neutron | Nucleus | Neutral (no charge) | ~1 (or 1 atomic mass unit) |
| Electron | Electron cloud (orbitals around the nucleus) | Negative | ~1/1836 (negligible compared to proton/neutron, often approximated as 0 for simplicity in basic models) |
of Subatomic Particle Properties:
- Proton: Found in the nucleus, has a positive charge, and a relative weight of approximately 1 (same as a neutron, much heavier than an electron).
- Neutron: Also found in the nucleus, has no charge (neutral), and a relative weight of approximately 1 (same as a proton).
- Electron: Orbits the nucleus in electron clouds (or orbitals), has a negative charge, and a very small relative weight (≈1/1836 of a proton/neutron).
For the "Atom Timeline" (matching atomic models to years):
The timeline at the bottom likely corresponds to historical atomic models:
- Early model (atom is tiny, hard, indivisible): Democritus (ancient, but if we use the given descriptions, this matches early "indivisible sphere" models).
- "Solid, indivisible sphere" (can’t destroy/reconstruct): Dalton’s atomic model (early 1800s, 1803).
- "Plum pudding model" (atom is a sphere of positive charge with electrons embedded): Thomson’s model (1897–1904).
- "Divisible units with positive charge in nucleus, electrons orbiting": Rutherford’s nuclear model (1911).
- "Electrons in circular orbits (like planets)": Bohr’s model (1913).
- "Electron cloud (probability of finding electrons)": Quantum mechanical model (1920s–1930s).
If you need help with specific blanks (like labeling the timeline or the oxygen atom’s parts), let me know! For example, the oxygen atom diagram has:
- The central cluster (nucleus) with protons and neutrons.
- Electrons orbiting (or in the electron cloud) around the nucleus.
- The atomic symbol \( \ce{O} \) with atomic number 8, symbol \( \ce{O} \), name "Oxygen", and atomic mass 15.999.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
of Subatomic Particle Properties:
- Proton: Found in the nucleus, has a positive charge, and a relative weight of approximately 1 (same as a neutron, much heavier than an electron).
- Neutron: Also found in the nucleus, has no charge (neutral), and a relative weight of approximately 1 (same as a proton).
- Electron: Orbits the nucleus in electron clouds (or orbitals), has a negative charge, and a very small relative weight (≈1/1836 of a proton/neutron).
For the "Atom Timeline" (matching atomic models to years):
The timeline at the bottom likely corresponds to historical atomic models:
- Early model (atom is tiny, hard, indivisible): Democritus (ancient, but if we use the given descriptions, this matches early "indivisible sphere" models).
- "Solid, indivisible sphere" (can’t destroy/reconstruct): Dalton’s atomic model (early 1800s, 1803).
- "Plum pudding model" (atom is a sphere of positive charge with electrons embedded): Thomson’s model (1897–1904).
- "Divisible units with positive charge in nucleus, electrons orbiting": Rutherford’s nuclear model (1911).
- "Electrons in circular orbits (like planets)": Bohr’s model (1913).
- "Electron cloud (probability of finding electrons)": Quantum mechanical model (1920s–1930s).
If you need help with specific blanks (like labeling the timeline or the oxygen atom’s parts), let me know! For example, the oxygen atom diagram has:
- The central cluster (nucleus) with protons and neutrons.
- Electrons orbiting (or in the electron cloud) around the nucleus.
- The atomic symbol \( \ce{O} \) with atomic number 8, symbol \( \ce{O} \), name "Oxygen", and atomic mass 15.999.