QUESTION IMAGE
Question
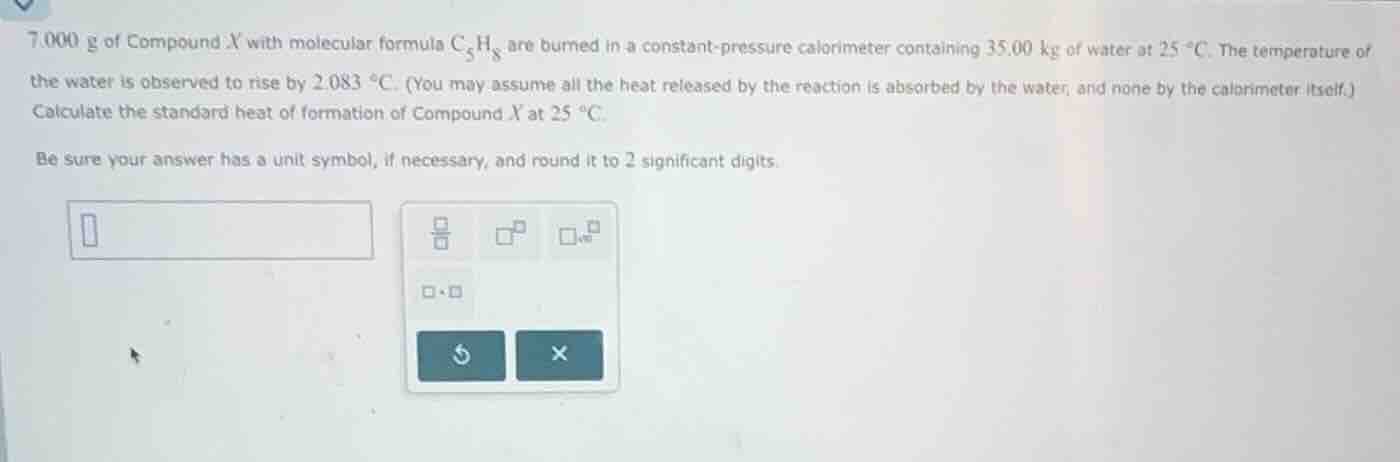
7.000 g of compound x with molecular formula \\(\ce{c5h8}\\) are burned in a constant - pressure calorimeter containing 35.00 kg of water at \\(25\\ ^{circ}\text{c}\\). the temperature of the water is observed to rise by \\(2.083\\ ^{circ}\text{c}\\). (you may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.) calculate the standard heat of formation of compound x at \\(25\\ ^{circ}\text{c}\\). be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.
Step1: Calculate heat absorbed by water
The formula for heat absorbed by water is \( q = mc\Delta T \), where \( m = 35.00\ kg = 35000\ g \), \( c = 4.184\ J/g^\circ C \), and \( \Delta T = 2.083^\circ C \).
\( q = 35000\ g\times4.184\ J/g^\circ C\times2.083^\circ C \)
\( q = 35000\times4.184\times2.083\ J \)
\( q \approx 35000\times8.716\ J \)
\( q \approx 3.0506\times10^{5}\ J = 305.06\ kJ \)
Step2: Calculate moles of \( C_5H_8 \)
Molar mass of \( C_5H_8 \): \( 5\times12.01 + 8\times1.008 = 60.05 + 8.064 = 68.114\ g/mol \)
Moles \( n = \frac{7.000\ g}{68.114\ g/mol} \approx 0.1028\ mol \)
Step3: Determine heat of combustion (\( \Delta H_{combustion} \))
Heat released by combustion is equal to heat absorbed by water (with sign: combustion is exothermic, so \( \Delta H_{combustion} = -\frac{q}{n} \))
\( \Delta H_{combustion} = -\frac{305.06\ kJ}{0.1028\ mol} \approx -2967\ kJ/mol \)
Step4: Write combustion reaction and use standard heats of formation
Combustion reaction: \( C_5H_8 + 7O_2
ightarrow 5CO_2 + 4H_2O(l) \)
Standard heats of formation: \( \Delta H_f(CO_2) = -393.5\ kJ/mol \), \( \Delta H_f(H_2O(l)) = -285.8\ kJ/mol \), \( \Delta H_f(O_2) = 0\ kJ/mol \)
\( \Delta H_{combustion} = \sum \Delta H_f(products) - \sum \Delta H_f(reactants) \)
\( -2967\ kJ/mol = [5\times(-393.5) + 4\times(-285.8)] - [\Delta H_f(C_5H_8) + 7\times0] \)
\( -2967 = [ -1967.5 - 1143.2 ] - \Delta H_f(C_5H_8) \)
\( -2967 = -3110.7 - \Delta H_f(C_5H_8) \)
\( \Delta H_f(C_5H_8) = -3110.7 + 2967 \)
\( \Delta H_f(C_5H_8) \approx -143.7\ kJ/mol \) (Wait, correction: Let's recalculate the sum of products)
Wait, recalculating \( 5\times(-393.5) = -1967.5 \), \( 4\times(-285.8) = -1143.2 \), sum: \( -1967.5 - 1143.2 = -3110.7 \)
So \( -2967 = -3110.7 - \Delta H_f(C_5H_8) \)
\( \Delta H_f(C_5H_8) = -3110.7 + 2967 = -143.7\ kJ/mol \)? Wait, no, sign error. Wait, \( \Delta H_{combustion} = \sum \Delta H_f(products) - \sum \Delta H_f(reactants) \)
So \( -2967 = (5\Delta H_f(CO_2) + 4\Delta H_f(H_2O)) - (\Delta H_f(C_5H_8) + 7\Delta H_f(O_2)) \)
Since \( \Delta H_f(O_2) = 0 \),
\( -2967 = (5\times(-393.5) + 4\times(-285.8)) - \Delta H_f(C_5H_8) \)
\( -2967 = (-1967.5 - 1143.2) - \Delta H_f(C_5H_8) \)
\( -2967 = -3110.7 - \Delta H_f(C_5H_8) \)
Then, \( \Delta H_f(C_5H_8) = -3110.7 + 2967 = -143.7\ kJ/mol \)? Wait, that can't be. Wait, maybe miscalculation in heat absorbed. Let's recalculate step1:
\( 35000\ g \times 4.184\ J/g^\circ C \times 2.083^\circ C \)
\( 35000\times4.184 = 146440 \)
\( 146440\times2.083 = 146440\times2 + 146440\times0.083 = 292880 + 12154.52 = 305034.52\ J = 305.03452\ kJ \)
Moles: \( 7.000 / 68.114 \approx 0.10277\ mol \)
\( \Delta H_{combustion} = -305.03452\ kJ / 0.10277\ mol \approx -2968\ kJ/mol \)
Now, reaction: \( C_5H_8 + 7O_2
ightarrow 5CO_2 + 4H_2O(l) \)
\( \Delta H_{combustion} = 5\Delta H_f(CO_2) + 4\Delta H_f(H_2O) - \Delta H_f(C_5H_8) - 7\Delta H_f(O_2) \)
\( -2968 = 5\times(-393.5) + 4\times(-285.8) - \Delta H_f(C_5H_8) - 0 \)
\( -2968 = -1967.5 - 1143.2 - \Delta H_f(C_5H_8) \)
\( -2968 = -3110.7 - \Delta H_f(C_5H_8) \)
\( \Delta H_f(C_5H_8) = -3110.7 + 2968 = -142.7\ kJ/mol \approx -1.4\times10^{2}\ kJ/mol \) (rounded to 2 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( -1.4\times10^{2}\ kJ/mol \) (or -140 kJ/mol, but more accurately -1.4×10² kJ/mol)