QUESTION IMAGE
Question
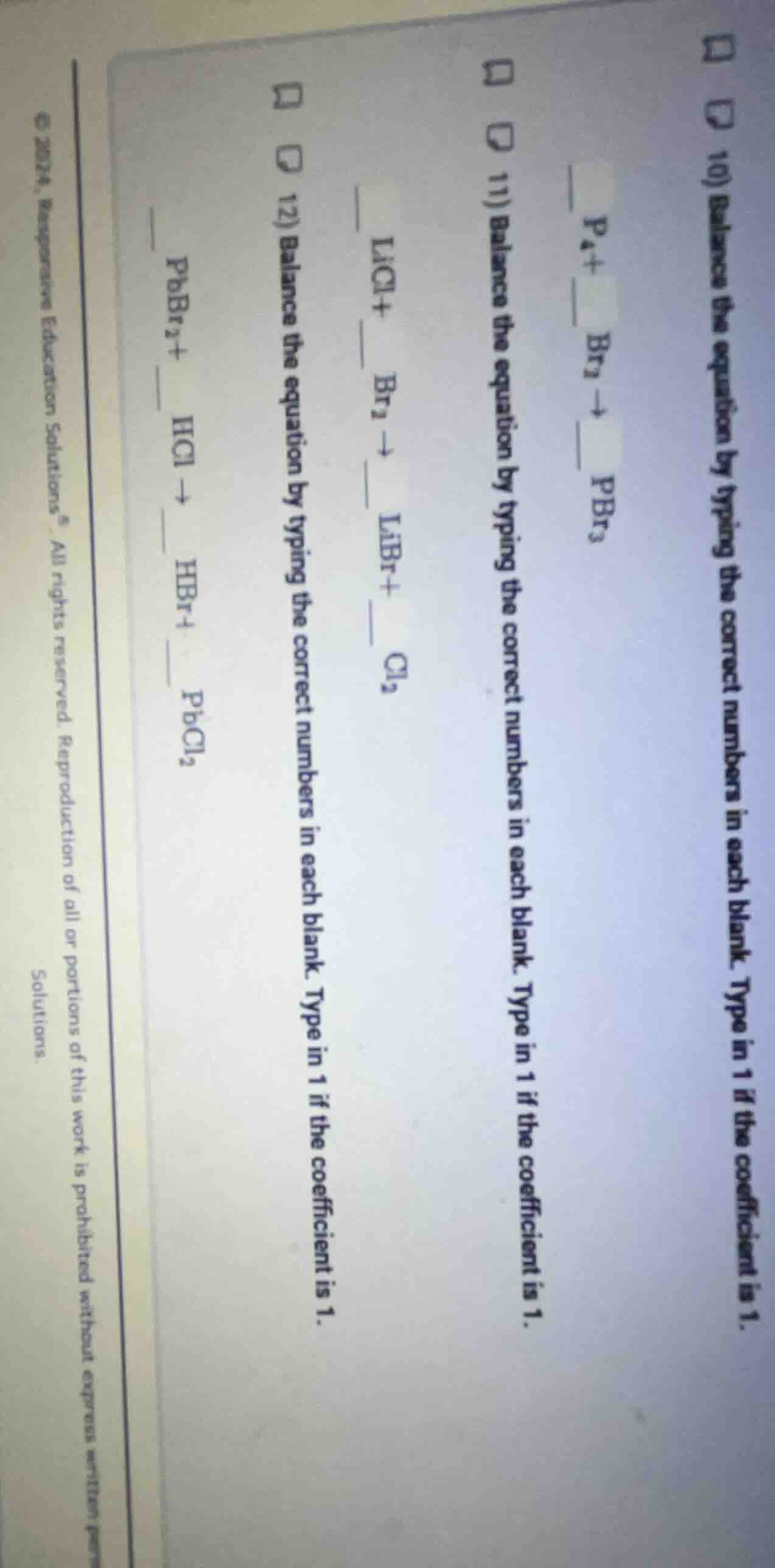
- balance the equation by typing the correct numbers in each blank. type in 1 if the coefficient is 1.
p₄ + br₂ → __ pbr₃
- balance the equation by typing the correct numbers in each blank. type in 1 if the coefficient is 1.
licl + br₂ → libr + cl₂
- balance the equation by typing the correct numbers in each blank. type in 1 if the coefficient is 1.
pbbr₂ + hcl → hbr + pbcl₂
Step1: Balance P atoms
$1\ \text{P}_4 + \underline{\quad}\ \text{Br}_2
ightarrow 4\ \text{PBr}_3$
Step2: Balance Br atoms
$1\ \text{P}_4 + 6\ \text{Br}_2
ightarrow 4\ \text{PBr}_3$
---
Step1: Balance Cl atoms
$2\ \text{LiCl} + \underline{\quad}\ \text{Br}_2
ightarrow \underline{\quad}\ \text{LiBr} + 1\ \text{Cl}_2$
Step2: Balance Li and Br atoms
$2\ \text{LiCl} + 1\ \text{Br}_2
ightarrow 2\ \text{LiBr} + 1\ \text{Cl}_2$
---
Step1: Balance Pb atoms
$1\ \text{PbBr}_2 + \underline{\quad}\ \text{HCl}
ightarrow \underline{\quad}\ \text{HBr} + 1\ \text{PbCl}_2$
Step2: Balance Cl and Br atoms
$1\ \text{PbBr}_2 + 2\ \text{HCl}
ightarrow 2\ \text{HBr} + 1\ \text{PbCl}_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $1\ \text{P}_4 + 6\ \text{Br}_2
ightarrow 4\ \text{PBr}_3$
- $2\ \text{LiCl} + 1\ \text{Br}_2
ightarrow 2\ \text{LiBr} + 1\ \text{Cl}_2$
- $1\ \text{PbBr}_2 + 2\ \text{HCl}
ightarrow 2\ \text{HBr} + 1\ \text{PbCl}_2$