QUESTION IMAGE
Question
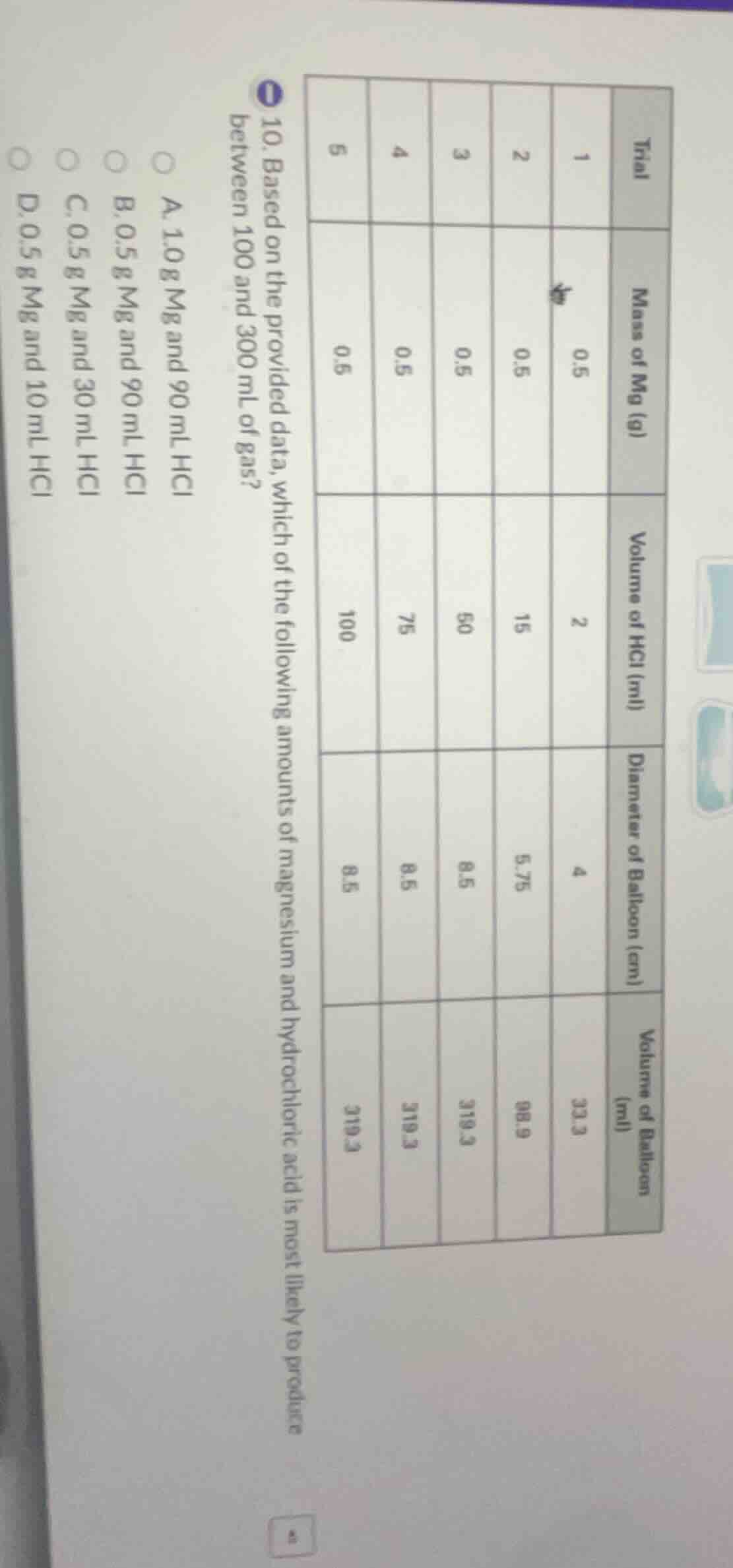
- based on the provided data, which of the following amounts of magnesium and hydrochloric acid is most likely to produce between 100 and 300 ml of gas? a. 1.0 g mg and 90 ml hcl b. 0.5 g mg and 90 ml hcl c. 0.5 g mg and 30 ml hcl d. 0.5 g mg and 10 ml hcl
Step1: Analyze data trends
From trials 1-5: 0.5g Mg with 2→15→50→75→100 mL HCl gives balloon volumes 33.3→98.9→319.3→319.3→319.3 mL. So 50+ mL HCl fully reacts 0.5g Mg to make ~319.3 mL gas.
Step2: Target gas range
We need 100-300 mL gas, so use less than full HCl for 0.5g Mg, or adjust Mg amount.
Step3: Evaluate options
- A: 1.0g Mg needs more HCl than 100 mL to fully react, gas would exceed 319.3×2=638.6 mL, too much.
- B: 0.5g Mg with 90 mL HCl: 90 mL ≥50 mL, so full reaction gives ~319.3 mL, over 300 mL.
- C: 0.5g Mg with 30 mL HCl: 30 mL <50 mL, so HCl is limiting. Gas volume is proportional to HCl: $\frac{30}{50} \times 319.3 = 191.6$ mL, which is 100-300 mL.
- D: 0.5g Mg with 10 mL HCl: $\frac{10}{50} \times 319.3 = 63.9$ mL, below 100 mL.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. 0.5 g Mg and 30 mL HCl