QUESTION IMAGE
Question
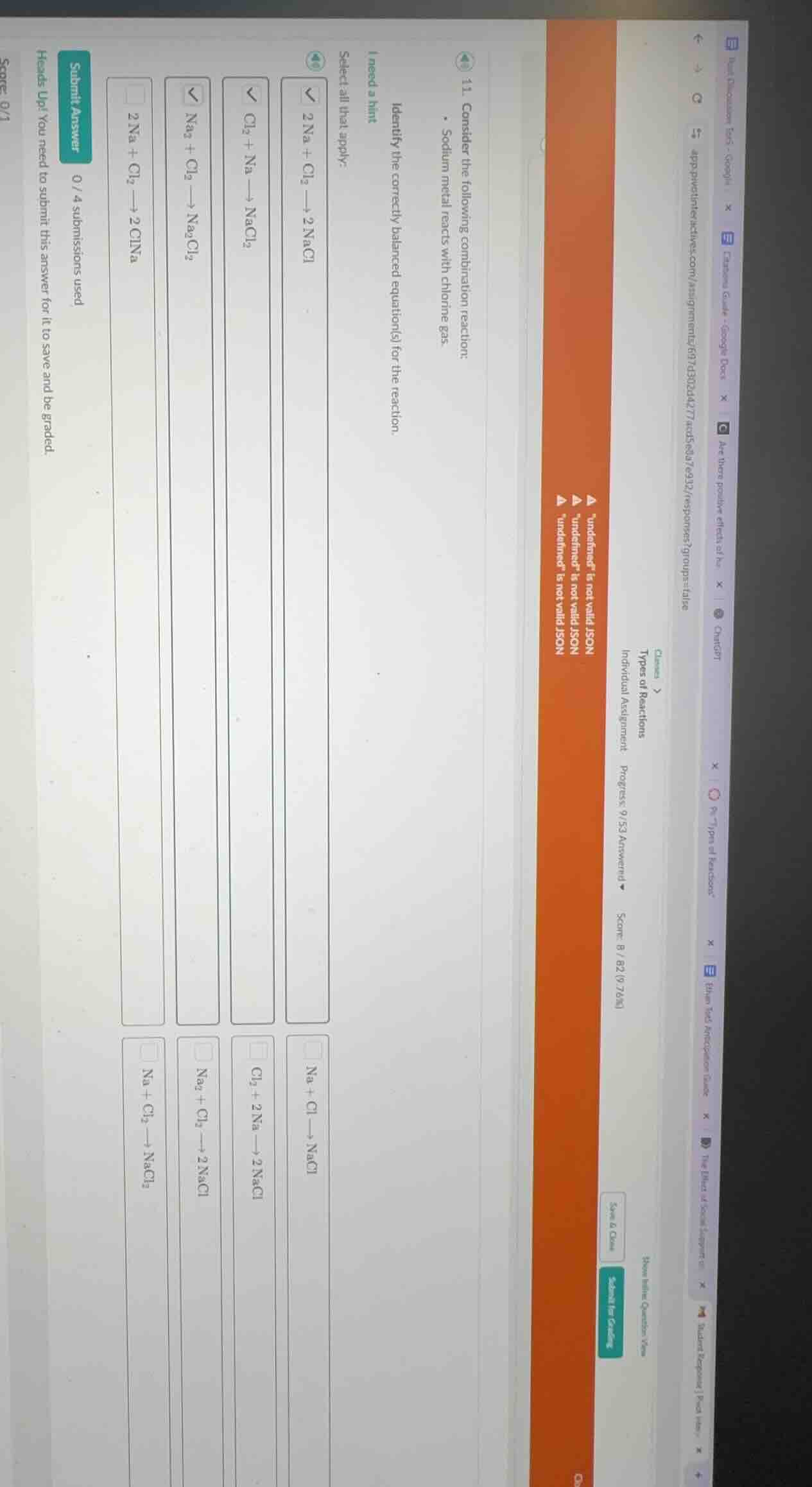
- consider the following combination reaction: sodium metal reacts with chlorine gas. identify the correctly balanced equation(s) for the reaction. select all that apply: 2 na + cl₂ → 2 nacl na + cl → nacl cl₂ + na → nacl₂ cl₂ + 2 na → 2 nacl na₂ + cl₂ → na₂cl₂ na₂ + cl₂ → 2 nacl 2 na + cl₂ → 2 clna na + cl₂ → nacl₂
Step1: Check Na + Cl → NaCl
Count atoms: Left: 1 Na, 1 Cl; Right:1 Na,1 Cl. But chlorine exists as $\text{Cl}_2$, so invalid.
Step2: Check $\text{Cl}_2 + 2\text{Na}
ightarrow 2\text{NaCl}$
Count atoms: Left:2 Na, 2 Cl; Right:2 Na,2 Cl. Correct formula/balance.
Step3: Check $\text{Na}_2 + \text{Cl}_2
ightarrow 2\text{NaCl}$
Sodium exists as single Na atoms, not $\text{Na}_2$, so invalid.
Step4: Check $\text{Na} + \text{Cl}_2
ightarrow \text{NaCl}_2$
Product $\text{NaCl}_2$ is invalid; correct product is NaCl, so invalid.
Step5: Check $2\text{Na} + \text{Cl}_2
ightarrow 2\text{NaCl}$
Count atoms: Left:2 Na, 2 Cl; Right:2 Na,2 Cl. Correct formula/balance.
Step6: Check $\text{Cl}_2 + \text{Na}
ightarrow \text{NaCl}_2$
Product $\text{NaCl}_2$ is invalid; unbalanced, so invalid.
Step7: Check $\text{Na}_2 + \text{Cl}_2
ightarrow \text{Na}_2\text{Cl}_2$
Sodium exists as single Na, product formula is wrong, so invalid.
Step8: Check $2\text{Na} + \text{Cl}_2
ightarrow 2\text{ClNa}$
Formula $\text{ClNa}$ is non-standard (correct is NaCl), so invalid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{2\text{Na} + \text{Cl}_2
ightarrow 2\text{NaCl}}$
$\boldsymbol{\text{Cl}_2 + 2\text{Na}
ightarrow 2\text{NaCl}}$