QUESTION IMAGE
Question
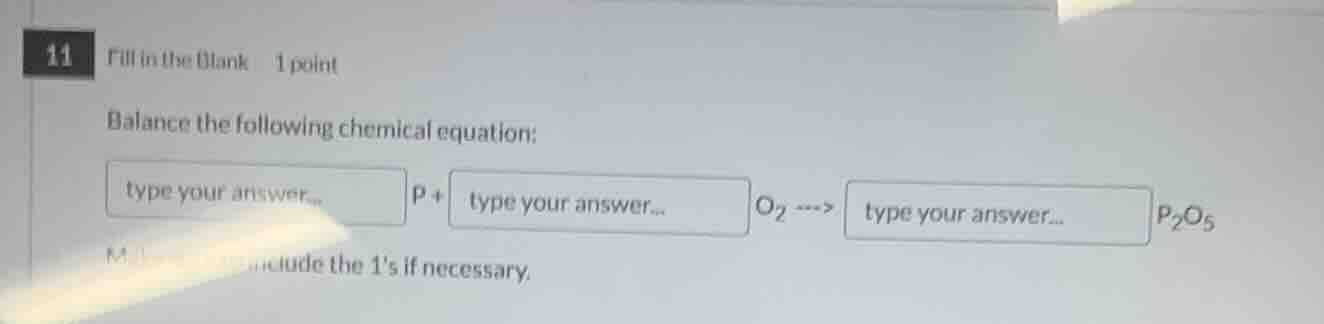
11 fill in the blank 1 point balance the following chemical equation: type your answer... p + type your answer... o₂ ---> type your answer... p₂o₅ include the 1s if necessary.
Step1: Balance phosphorus atoms
Count P on right: 2 in $\text{P}_2\text{O}_5$. Add coefficient 2 to left P:
$2\text{P} + \text{O}_2
ightarrow \text{P}_2\text{O}_5$
Step2: Balance oxygen atoms
Count O on right: 5 in $\text{P}_2\text{O}_5$. Use $\frac{5}{2}$ for $\text{O}_2$:
$2\text{P} + \frac{5}{2}\text{O}_2
ightarrow \text{P}_2\text{O}_5$
Step3: Eliminate fractional coefficient
Multiply all terms by 2:
$4\text{P} + 5\text{O}_2
ightarrow 2\text{P}_2\text{O}_5$
Step4: Verify atom counts
Left: 4 P, 10 O; Right: 4 P, 10 O. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
4, 5, 2