QUESTION IMAGE
Question
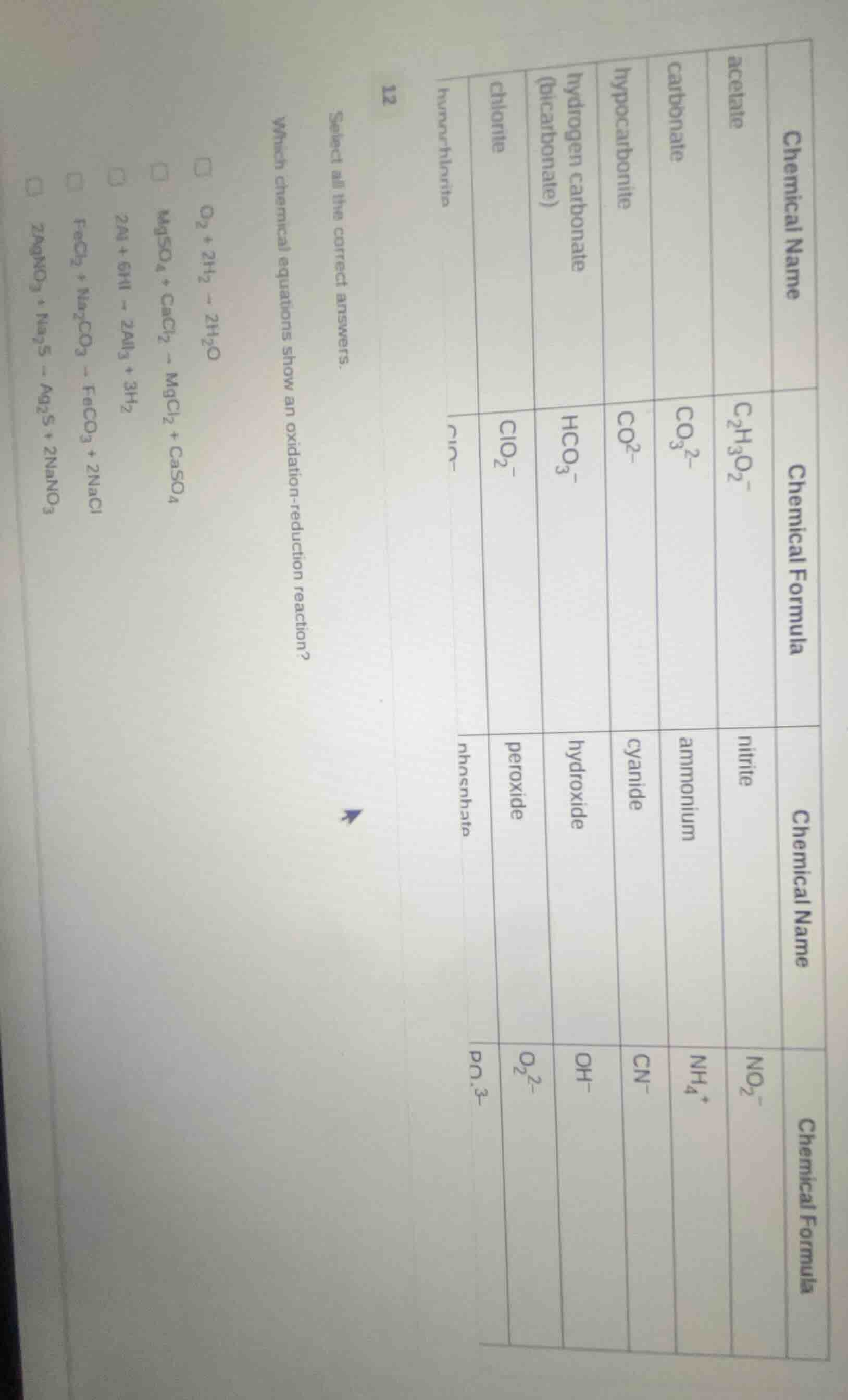
12
select all the correct answers.
which chemical equations show an oxidation - reduction reaction?
□ \\(\ce{o_{2} + 2h_{2} -> 2h_{2}o}\\)
□ \\(\ce{mgso_{4} + cacl_{2} -> mgcl_{2} + caso_{4}}\\)
□ \\(\ce{2al + 6hi -> 2ali_{3} + 3h_{2}}\\)
□ \\(\ce{fecl_{2} + na_{2}co_{3} -> feco_{3} + 2nacl}\\)
□ \\(\ce{2agno_{3} + na_{2}s -> ag_{2}s + 2nano_{3}}\\)
| chemical name | chemical formula | chemical name | chemical formula |
|---|---|---|---|
| carbonate | \\(\ce{co_{3}^{2 -}}\\) | ammonium | \\(\ce{nh_{4}^{+}}\\) |
| hypocarbonite | \\(\ce{co_{2}^{2 -}}\\) | cyanide | \\(\ce{cn^{-}}\\) |
| hydrogen carbonate (bicarbonate) | \\(\ce{hco_{3}^{-}}\\) | hydroxide | \\(\ce{oh^{-}}\\) |
| chlorite | \\(\ce{clo_{2}^{-}}\\) | peroxide | \\(\ce{o_{2}^{2 -}}\\) |
| hypochlorite | \\(\ce{clo^{-}}\\) | phosphate | \\(\ce{po_{4}^{3 -}}\\) |
To determine which chemical equations represent oxidation - reduction (redox) reactions, we need to check for changes in oxidation states of the elements involved. A redox reaction is defined as a reaction where there is a transfer of electrons, which is manifested as a change in the oxidation number of one or more elements.
Analyzing Equation 1: $\boldsymbol{O_2 + 2H_2
ightarrow 2H_2O}$
- For oxygen ($O_2$), the oxidation state of $O$ in $O_2$ (elemental form) is $0$. In $H_2O$, the oxidation state of $O$ is $ - 2$. So, oxygen is reduced (its oxidation state decreases).
- For hydrogen ($H_2$), the oxidation state of $H$ in $H_2$ (elemental form) is $0$. In $H_2O$, the oxidation state of $H$ is $+ 1$. So, hydrogen is oxidized (its oxidation state increases).
- Since there are changes in oxidation states for both $O$ and $H$, this is a redox reaction.
Analyzing Equation 2: $\boldsymbol{MgSO_4+CaCl_2
ightarrow MgCl_2 + CaSO_4}$
- Let's find the oxidation states of each element:
- In $MgSO_4$: $Mg$ has an oxidation state of $+ 2$, $S$ has an oxidation state of $+ 6$, and $O$ has an oxidation state of $ - 2$.
- In $CaCl_2$: $Ca$ has an oxidation state of $+ 2$, and $Cl$ has an oxidation state of $ - 1$.
- In $MgCl_2$: $Mg$ has an oxidation state of $+ 2$, and $Cl$ has an oxidation state of $ - 1$.
- In $CaSO_4$: $Ca$ has an oxidation state of $+ 2$, $S$ has an oxidation state of $+ 6$, and $O$ has an oxidation state of $ - 2$.
- There is no change in the oxidation states of any of the elements ($Mg$, $S$, $O$, $Ca$, $Cl$) during this reaction. This is a double - displacement (metathesis) reaction, not a redox reaction.
Analyzing Equation 3: $\boldsymbol{2Al + 6HI
ightarrow 2AlI_3+3H_2}$
- For aluminum ($Al$), the oxidation state of $Al$ in its elemental form is $0$. In $AlI_3$, the oxidation state of $Al$ is $+ 3$. So, aluminum is oxidized.
- For hydrogen ($H$), in $HI$, the oxidation state of $H$ is $+ 1$. In $H_2$ (elemental form), the oxidation state of $H$ is $0$. So, hydrogen is reduced.
- Since there are changes in oxidation states for $Al$ (oxidized) and $H$ (reduced), this is a redox reaction.
Analyzing Equation 4: $\boldsymbol{FeCl_2+Na_2CO_3
ightarrow FeCO_3+2NaCl}$
- Let's determine the oxidation states:
- In $FeCl_2$: $Fe$ has an oxidation state of $+ 2$, and $Cl$ has an oxidation state of $ - 1$.
- In $Na_2CO_3$: $Na$ has an oxidation state of $+ 1$, $C$ has an oxidation state of $+ 4$, and $O$ has an oxidation state of $ - 2$.
- In $FeCO_3$: $Fe$ has an oxidation state of $+ 2$, $C$ has an oxidation state of $+ 4$, and $O$ has an oxidation state of $ - 2$.
- In $NaCl$: $Na$ has an oxidation state of $+ 1$, and $Cl$ has an oxidation state of $ - 1$.
- There is no change in the oxidation states of any of the elements ($Fe$, $Cl$, $Na$, $C$, $O$) during this reaction. This is a double - displacement reaction, not a redox reaction.
Analyzing Equation 5: $\boldsymbol{2AgNO_3+Na_2S
ightarrow Ag_2S + 2NaNO_3}$
- Let's check the oxidation states:
- In $AgNO_3$: $Ag$ has an oxidation state of $+ 1$, $N$ has an oxidation state of $+ 5$, and $O$ has an oxidation state of $ - 2$.
- In $Na_2S$: $Na$ has an oxidation state of $+ 1$, and $S$ has an oxidation state of $ - 2$.
- In $Ag_2S$: $Ag$ has an oxidation state of $+ 1$, and $S$ has an oxidation state of $ - 2$.
- In $NaNO_3$: $Na$ has an oxidation state of $+ 1$, $N$ has an oxidation state of $+ 5$, and $O$ has an oxidation state of $ - 2$.
- There is no change in the oxidation states of any of the elements ($A…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The chemical equations that show an oxidation - reduction reaction are $\boldsymbol{O_2 + 2H_2
ightarrow 2H_2O}$ and $\boldsymbol{2Al + 6HI
ightarrow 2AlI_3+3H_2}$.