QUESTION IMAGE
Question
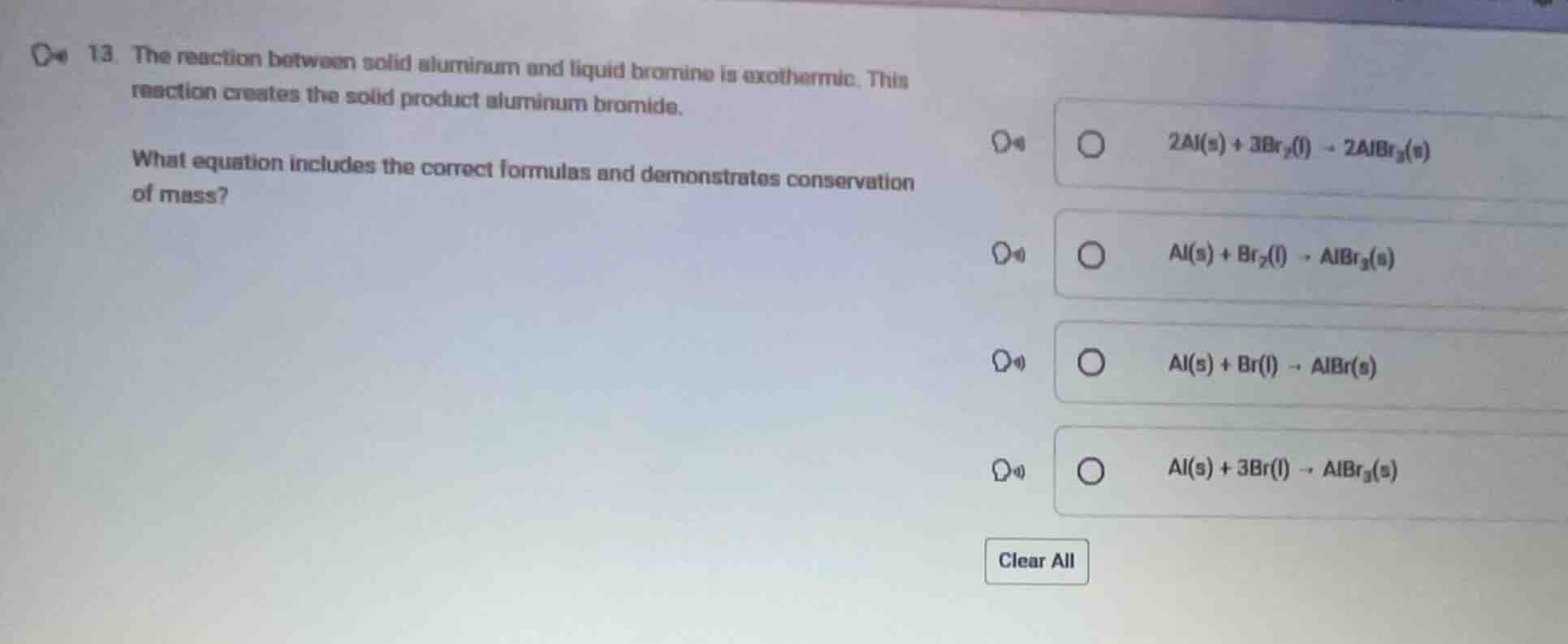
- the reaction between solid aluminum and liquid bromine is exothermic. this reaction creates the solid product aluminum bromide. what equation includes the correct formulas and demonstrates conservation of mass? \\( 2\text{al}(s) + 3\text{br}_2(l) \
ightarrow 2\text{albr}_3(s) \\) \\( \text{al}(s) + \text{br}_2(l) \
ightarrow \text{albr}_3(s) \\) \\( \text{al}(s) + \text{br}(l) \
ightarrow \text{albr}(s) \\) \\( \text{al}(s) + 3\text{br}(l) \
ightarrow \text{albr}_3(s) \\)
Step1: Identify correct formulas
Aluminum is $\text{Al}(s)$, bromine exists as diatomic liquid $\text{Br}_2(l)$, aluminum bromide is $\text{AlBr}_3(s)$ (Al has +3 charge, Br has -1 charge, so 3 Br⁻ balance 1 Al³⁺). This eliminates options C and D (wrong bromine formula).
Step2: Balance the equation
Count atoms on each side:
- Unbalanced: $\text{Al}(s) + \text{Br}_2(l)
ightarrow \text{AlBr}_3(s)$
- Balance Br: 3 $\text{Br}_2$ gives 6 Br, so need 2 $\text{AlBr}_3$ (6 Br total).
- Balance Al: 2 $\text{Al}$ to match 2 Al in $2\text{AlBr}_3$.
Final balanced equation: $2\text{Al}(s) + 3\text{Br}_2(l)
ightarrow 2\text{AlBr}_3(s)$, which matches option A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $2\text{Al}(s) + 3\text{Br}_2(l)
ightarrow 2\text{AlBr}_3(s)$