QUESTION IMAGE
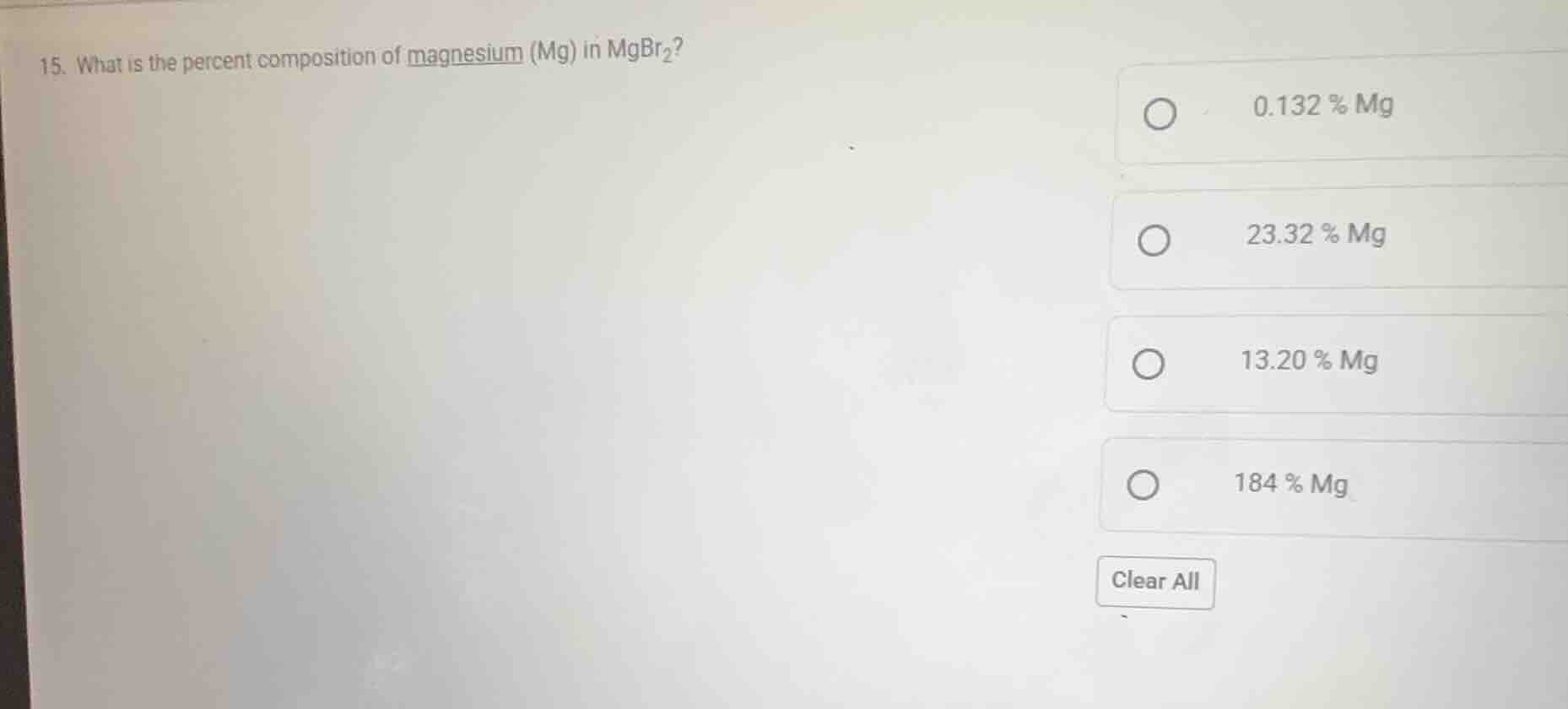
Question
- what is the percent composition of magnesium (mg) in mgbr₂?
0.132 % mg
23.32 % mg
13.20 % mg
184 % mg
clear all
Step1: Find molar masses
Molar mass of Mg: $24.31\ g/mol$.
Molar mass of Br: $79.90\ g/mol$, so for $Br_2$: $2\times79.90 = 159.80\ g/mol$.
Step2: Calculate molar mass of $MgBr_2$
Molar mass of $MgBr_2$ = $24.31 + 159.80 = 184.11\ g/mol$.
Step3: Calculate percent composition of Mg
Percent composition = $\frac{\text{Molar mass of Mg}}{\text{Molar mass of } MgBr_2} \times 100\%$
= $\frac{24.31}{184.11} \times 100\% \approx 13.20\%$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
13.20 % Mg (corresponding to the option "13.20 % Mg")