QUESTION IMAGE
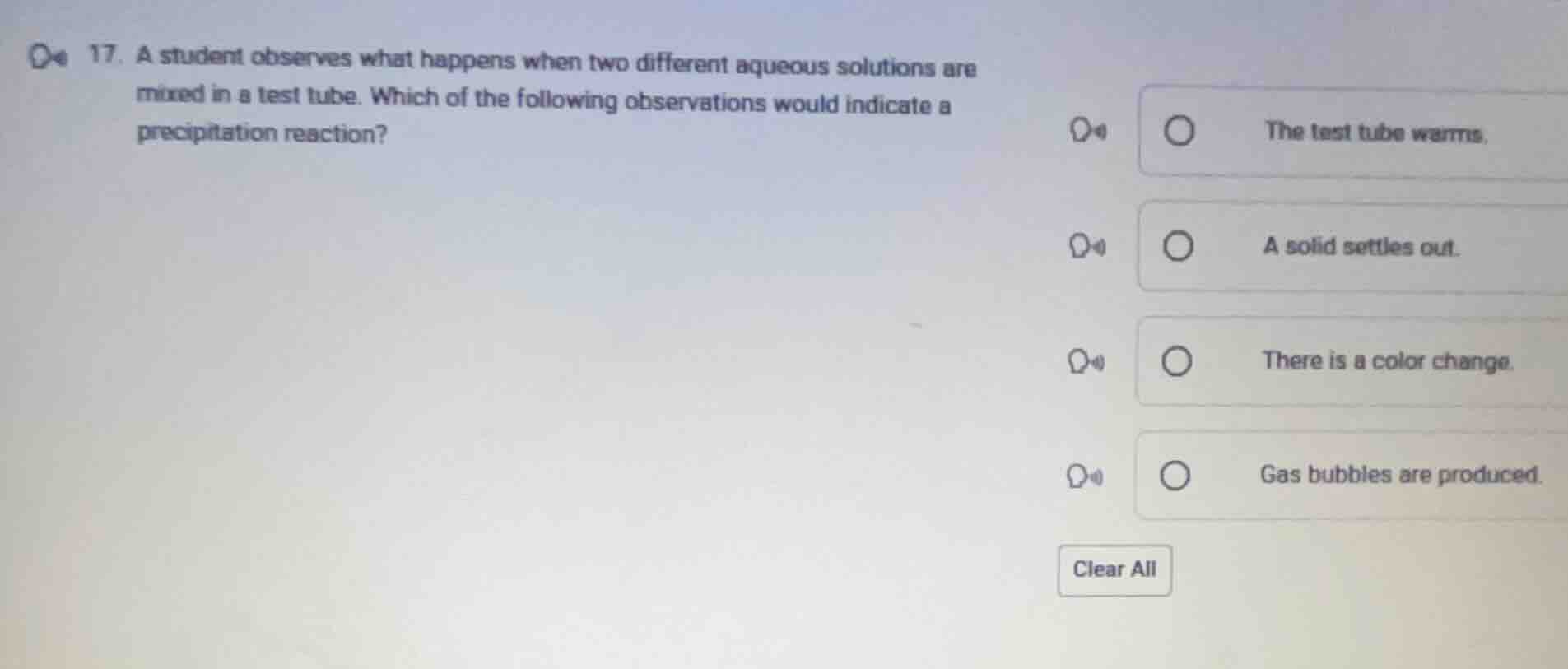
Question
- a student observes what happens when two different aqueous solutions are mixed in a test tube. which of the following observations would indicate a precipitation reaction? the test tube warms. a solid settles out. there is a color change. gas bubbles are produced. clear all
Brief Explanations
A precipitation reaction is defined as a chemical reaction where an insoluble solid (precipitate) forms from mixing aqueous solutions. A warming test tube indicates an exothermic reaction, a color change can occur in various reaction types, and gas bubbles signal a gas-producing reaction, while a settling solid is the defining observation for precipitation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. A solid settles out.