QUESTION IMAGE
Question
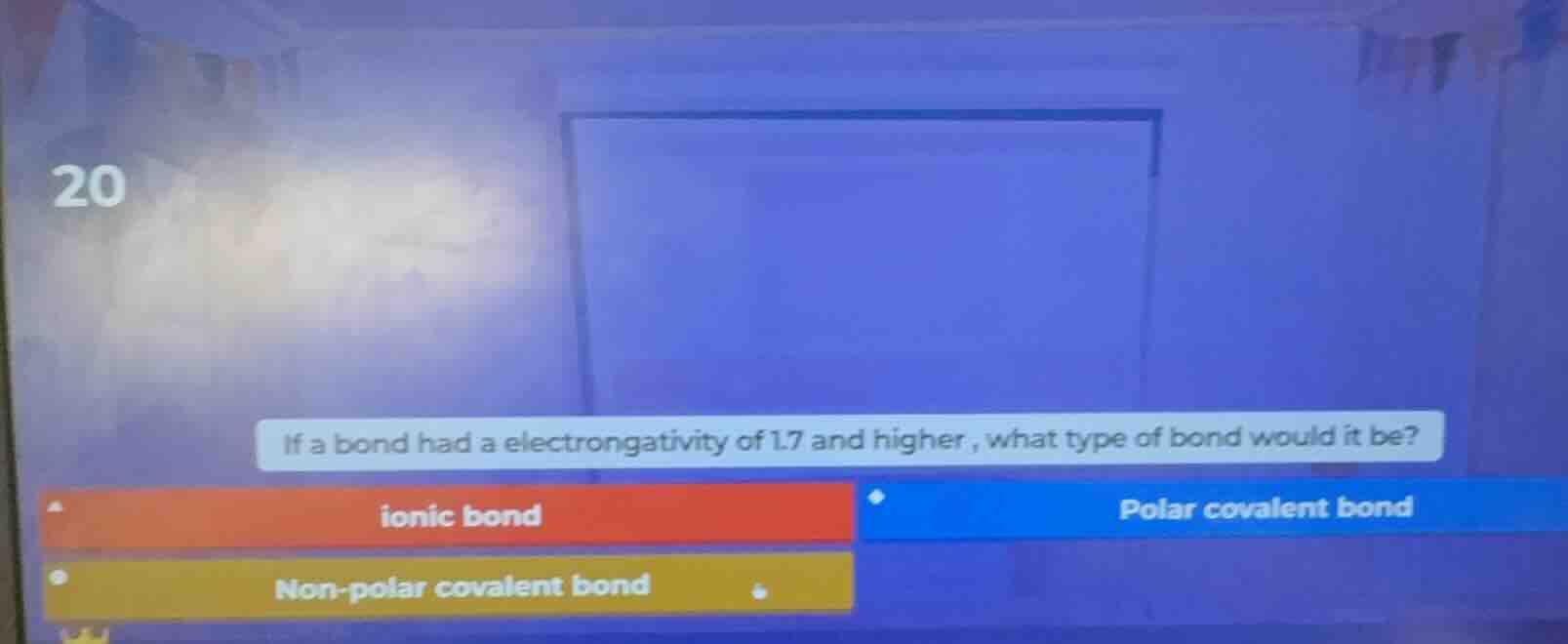
20
if a bond had a electrongativity of 1.7 and higher, what type of bond would it be?
ionic bond
polar covalent bond
non-polar covalent bond
Brief Explanations
Electronegativity difference is used to classify bonds. A difference of 1.7 or higher is the standard threshold for an ionic bond, where electrons are transferred rather than shared. Polar covalent bonds have smaller differences (0.4 to 1.7), and non-polar covalent bonds have differences below 0.4.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. ionic bond