QUESTION IMAGE
Question
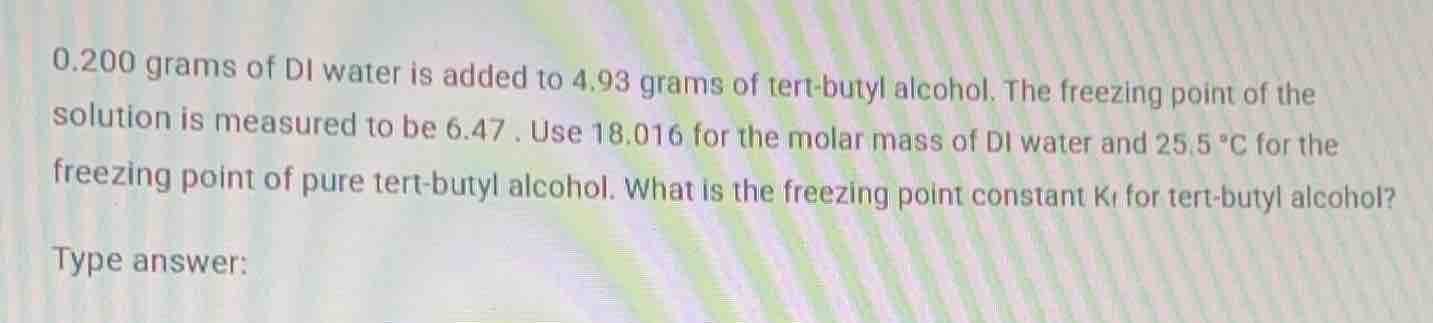
0.200 grams of di water is added to 4.93 grams of tert - butyl alcohol. the freezing point of the solution is measured to be 6.47. use 18.016 for the molar mass of di water and 25.5 °c for the freezing point of pure tert - butyl alcohol. what is the freezing point constant ( k_f ) for tert - butyl alcohol? type answer:
Step1: Calculate moles of water (solute)
Moles of water = mass / molar mass = \( \frac{0.200\ g}{18.016\ g/mol} \approx 0.0111\ mol \)
Step2: Calculate mass of solvent (tert - butyl alcohol) in kg
Mass of solvent = \( 4.93\ g = 4.93\times10^{- 3}\ kg \)
Step3: Calculate molality (m) of the solution
Molality \( m=\frac{moles\ of\ solute}{mass\ of\ solvent\ (kg)}=\frac{0.0111\ mol}{4.93\times10^{-3}\ kg}\approx2.252\ m \)
Step4: Calculate freezing point depression (\(\Delta T_f\))
\(\Delta T_f = T_{f,pure}-T_{f,solution}=25.5^{\circ}C - 6.47^{\circ}C = 19.03^{\circ}C\)
Step5: Use freezing point depression formula \(\Delta T_f = K_f\times m\) to find \(K_f\)
\(K_f=\frac{\Delta T_f}{m}=\frac{19.03^{\circ}C}{2.252\ m}\approx8.45^{\circ}C/m\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(8.45\ ^{\circ}\text{C}/m\) (or a value close to it depending on more precise calculations)