QUESTION IMAGE
Question
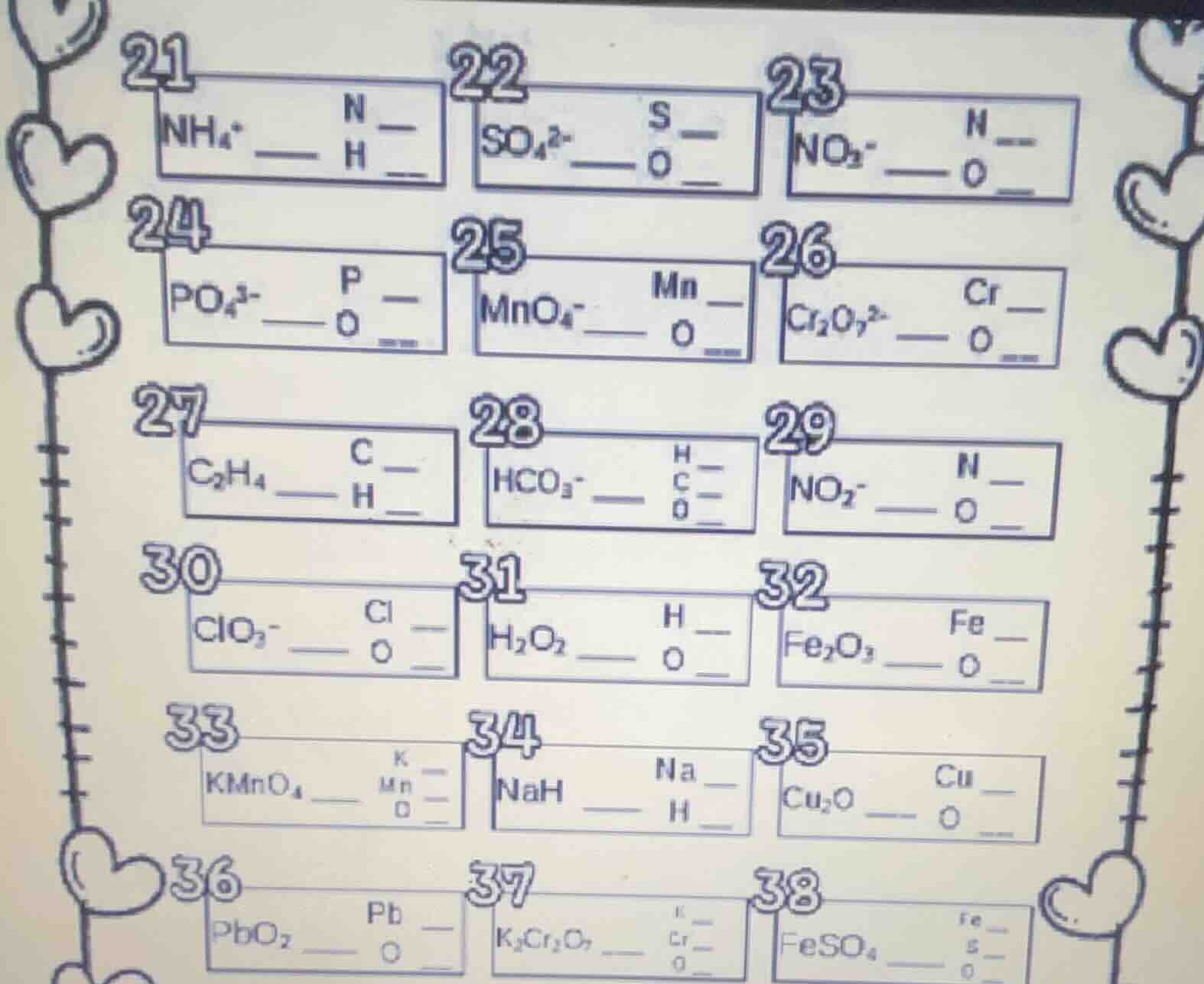
21 nh₄⁺ —— n h
22 so₄²⁻ —— s o
23 no₂⁻ —— n o
24 po₄³⁻ —— p o
25 mno₄⁻ —— mn o
26 cr₂o₇²⁻ —— cr o
27 c₂h₄ —— c h
28 hco₃⁻ —— h c o __
29 no₂⁻ —— n o
30 clo₃⁻ —— cl o
31 h₂o₂ —— h o
32 fe₂o₃ —— fe o
33 kmno₄ —— k mn o __
34 nah —— na h
35 cu₂o —— cu o
36 pbo₂ —— pb o
37 k₂cr₂o₇ —— k cr o __
38 feso₄ —— fe s o __
To solve for the oxidation states of the elements in each compound/ion, we use the rule that the sum of oxidation states in a neutral compound is 0, and in a polyatomic ion is equal to the ion's charge. Here are the solutions for a few examples (let's take problem 21, 22, and 32 as samples):
Problem 21: $\boldsymbol{NH_4^+}$
Step 1: Let the oxidation state of $N$ be $x$, and $H$ is usually $+1$ (in compounds with non - metals).
The sum of oxidation states in $NH_4^+$ (charge = $+1$) is given by the equation: $x + 4\times(+1)=+1$.
Step 2: Solve for $x$.
$x+4 = 1\implies x=1 - 4=-3$.
For $H$: We already used $+1$ in the calculation, and since the sum of $H$ oxidation states is $4\times(+1) = + 4$, each $H$ has an oxidation state of $+1$.
Problem 22: $\boldsymbol{SO_4^{2 - }}$
Step 1: Let the oxidation state of $S$ be $x$, and $O$ is usually $-2$ (in most compounds).
The sum of oxidation states in $SO_4^{2 - }$ (charge = $-2$) is: $x+4\times(-2)=-2$.
Step 2: Solve for $x$.
$x-8=-2\implies x=-2 + 8 = + 6$.
For $O$: We used $-2$ in the calculation, so each $O$ has an oxidation state of $-2$.
Problem 32: $\boldsymbol{Fe_2O_3}$
Step 1: Let the oxidation state of $Fe$ be $x$, and $O$ is $-2$.
The sum of oxidation states in $Fe_2O_3$ (neutral, so sum = 0) is: $2x+3\times(-2)=0$.
Step 2: Solve for $x$.
$2x-6 = 0\implies2x=6\implies x = + 3$.
For $O$: Each $O$ has an oxidation state of $-2$.
General Approach for Other Problems:
For each compound/ion:
- Identify the common oxidation state of the more electronegative element (e.g., $O$ is often $-2$, $H$ is often $+1$ in compounds with non - metals, $Na, K$ are $+1$, etc.).
- Let the oxidation state of the element in question be $x$.
- Set up an equation based on the sum of oxidation states (equal to the ion's charge or 0 for neutral compounds).
- Solve for $x$.
Example Answers for a Few More:
- Problem 23 ($NO_2^-$): Let $N$ be $x$, $O=-2$. Equation: $x + 2\times(-2)=-1\implies x-4=-1\implies x = + 3$; $O=-2$.
- Problem 25 ($MnO_4^-$): Let $Mn$ be $x$, $O=-2$. Equation: $x + 4\times(-2)=-1\implies x-8=-1\implies x = + 7$; $O=-2$.
- Problem 27 ($C_2H_4$): Neutral compound. Let $C$ be $x$, $H = + 1$. Equation: $2x+4\times(+1)=0\implies2x=-4\implies x=-2$; $H = + 1$.
If you want solutions for other specific problems (like 24, 26, etc.), you can apply the same steps: identify known oxidation states, set up the equation, and solve for the unknown.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve for the oxidation states of the elements in each compound/ion, we use the rule that the sum of oxidation states in a neutral compound is 0, and in a polyatomic ion is equal to the ion's charge. Here are the solutions for a few examples (let's take problem 21, 22, and 32 as samples):
Problem 21: $\boldsymbol{NH_4^+}$
Step 1: Let the oxidation state of $N$ be $x$, and $H$ is usually $+1$ (in compounds with non - metals).
The sum of oxidation states in $NH_4^+$ (charge = $+1$) is given by the equation: $x + 4\times(+1)=+1$.
Step 2: Solve for $x$.
$x+4 = 1\implies x=1 - 4=-3$.
For $H$: We already used $+1$ in the calculation, and since the sum of $H$ oxidation states is $4\times(+1) = + 4$, each $H$ has an oxidation state of $+1$.
Problem 22: $\boldsymbol{SO_4^{2 - }}$
Step 1: Let the oxidation state of $S$ be $x$, and $O$ is usually $-2$ (in most compounds).
The sum of oxidation states in $SO_4^{2 - }$ (charge = $-2$) is: $x+4\times(-2)=-2$.
Step 2: Solve for $x$.
$x-8=-2\implies x=-2 + 8 = + 6$.
For $O$: We used $-2$ in the calculation, so each $O$ has an oxidation state of $-2$.
Problem 32: $\boldsymbol{Fe_2O_3}$
Step 1: Let the oxidation state of $Fe$ be $x$, and $O$ is $-2$.
The sum of oxidation states in $Fe_2O_3$ (neutral, so sum = 0) is: $2x+3\times(-2)=0$.
Step 2: Solve for $x$.
$2x-6 = 0\implies2x=6\implies x = + 3$.
For $O$: Each $O$ has an oxidation state of $-2$.
General Approach for Other Problems:
For each compound/ion:
- Identify the common oxidation state of the more electronegative element (e.g., $O$ is often $-2$, $H$ is often $+1$ in compounds with non - metals, $Na, K$ are $+1$, etc.).
- Let the oxidation state of the element in question be $x$.
- Set up an equation based on the sum of oxidation states (equal to the ion's charge or 0 for neutral compounds).
- Solve for $x$.
Example Answers for a Few More:
- Problem 23 ($NO_2^-$): Let $N$ be $x$, $O=-2$. Equation: $x + 2\times(-2)=-1\implies x-4=-1\implies x = + 3$; $O=-2$.
- Problem 25 ($MnO_4^-$): Let $Mn$ be $x$, $O=-2$. Equation: $x + 4\times(-2)=-1\implies x-8=-1\implies x = + 7$; $O=-2$.
- Problem 27 ($C_2H_4$): Neutral compound. Let $C$ be $x$, $H = + 1$. Equation: $2x+4\times(+1)=0\implies2x=-4\implies x=-2$; $H = + 1$.
If you want solutions for other specific problems (like 24, 26, etc.), you can apply the same steps: identify known oxidation states, set up the equation, and solve for the unknown.