QUESTION IMAGE
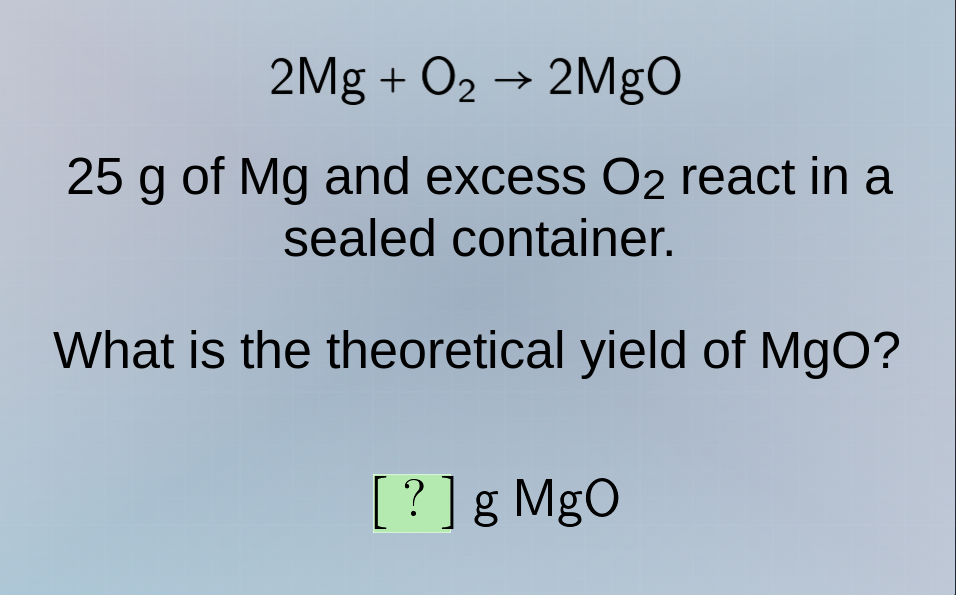
Question
2mg + o₂ → 2mgo
25 g of mg and excess o₂ react in a sealed container.
what is the theoretical yield of mgo?
? g mgo
Step1: Calculate moles of Mg
Molar mass of Mg is $24.31\ \text{g/mol}$. Moles of Mg = $\frac{\text{mass of Mg}}{\text{molar mass of Mg}}$ = $\frac{25\ \text{g}}{24.31\ \text{g/mol}} \approx 1.028\ \text{mol}$.
Step2: Relate moles of Mg to MgO
From the reaction $2\text{Mg} + \text{O}_2
ightarrow 2\text{MgO}$, the mole ratio of $\text{Mg}$ to $\text{MgO}$ is $2:2$ (or $1:1$). So moles of $\text{MgO}$ = moles of $\text{Mg}$ $\approx 1.028\ \text{mol}$.
Step3: Calculate mass of MgO
Molar mass of $\text{MgO}$ is $24.31 + 16.00 = 40.31\ \text{g/mol}$. Mass of $\text{MgO}$ = moles $\times$ molar mass = $1.028\ \text{mol} \times 40.31\ \text{g/mol} \approx 41.4\ \text{g}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\approx 41.4$