QUESTION IMAGE
Question
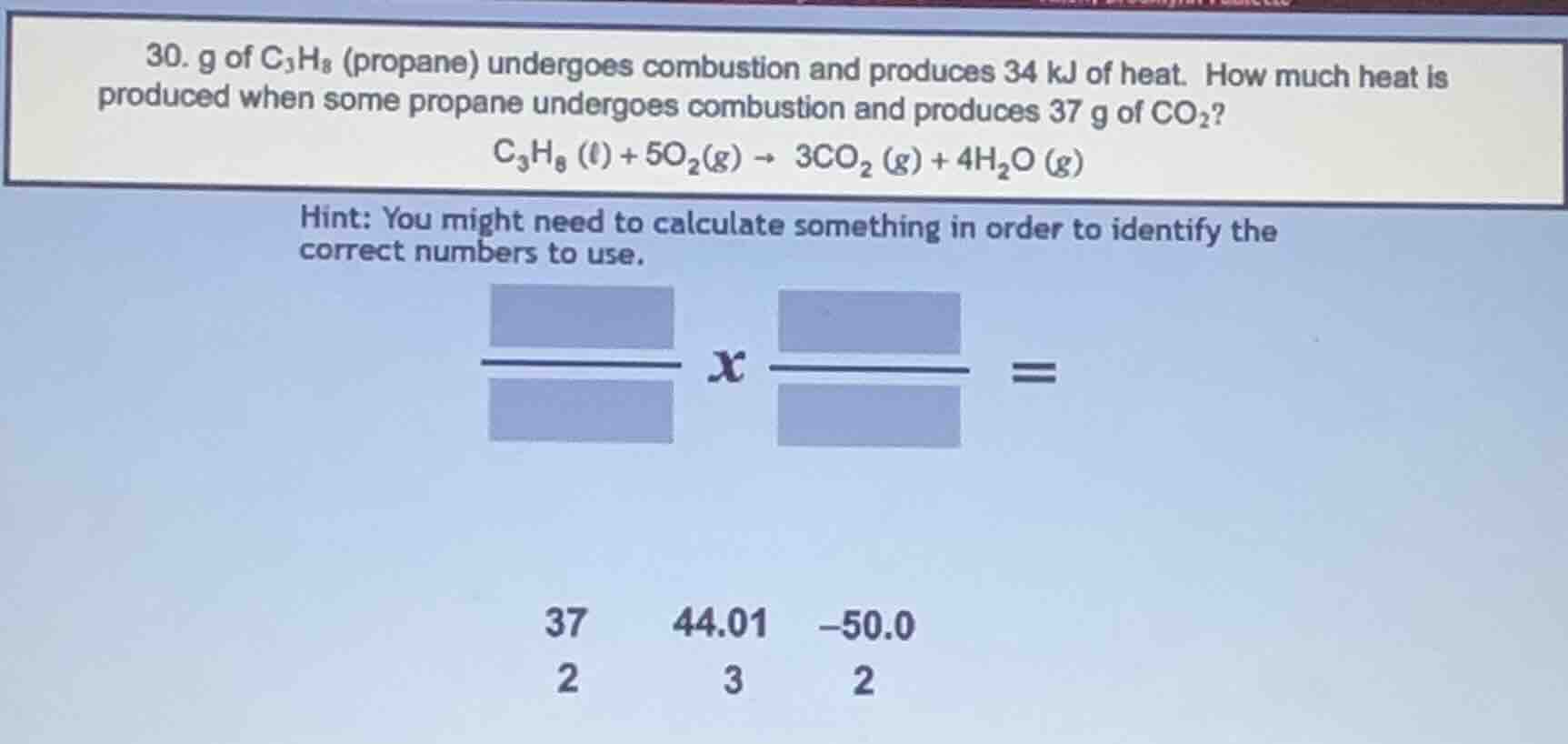
- g of c₃h₈ (propane) undergoes combustion and produces 34 kj of heat. how much heat is produced when some propane undergoes combustion and produces 37 g of co₂?
c₃h₈ (ℓ) + 5o₂(g) → 3co₂ (g) + 4h₂o (g)
hint: you might need to calculate something in order to identify the correct numbers to use.
blue rectangles and numbers 37, 44.01, -50.0, 2, 3, 2 as shown in the image
Step1: Find molar mass of $\text{C}_3\text{H}_8$
Molar mass of $\text{C}_3\text{H}_8 = 3\times12.01 + 8\times1.008 = 44.094\ \text{g/mol}$
Step2: Moles of $\text{C}_3\text{H}_8$ burned
$\text{Moles of } \text{C}_3\text{H}_8 = \frac{30.\ \text{g}}{44.094\ \text{g/mol}} \approx 0.680\ \text{mol}$
Step3: Heat per mole of $\text{C}_3\text{H}_8$
$\text{Heat per mole} = \frac{34\ \text{kJ}}{0.680\ \text{mol}} = 50.0\ \text{kJ/mol}$ (heat released, so $\Delta H = -50.0\ \text{kJ/mol}$)
Step4: Moles of $\text{CO}_2$ produced
Molar mass of $\text{CO}_2 = 44.01\ \text{g/mol}$
$\text{Moles of } \text{CO}_2 = \frac{37\ \text{g}}{44.01\ \text{g/mol}} \approx 0.841\ \text{mol}$
Step5: Moles of $\text{C}_3\text{H}_8$ for $\text{CO}_2$
From reaction: 1 mol $\text{C}_3\text{H}_8$ → 3 mol $\text{CO}_2$
$\text{Moles of } \text{C}_3\text{H}_8 = 0.841\ \text{mol CO}_2 \times \frac{1\ \text{mol C}_3\text{H}_8}{3\ \text{mol CO}_2} \approx 0.280\ \text{mol}$
Step6: Calculate total heat produced
$\text{Heat} = 0.280\ \text{mol} \times 50.0\ \text{kJ/mol} = 14\ \text{kJ}$
(Using the given template structure: $\frac{37}{44.01} \times \frac{-50.0}{3} \approx -14\ \text{kJ}$, negative sign indicates heat released)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
14 kJ of heat is produced (or -14 kJ if sign denotes heat release)