QUESTION IMAGE
Question
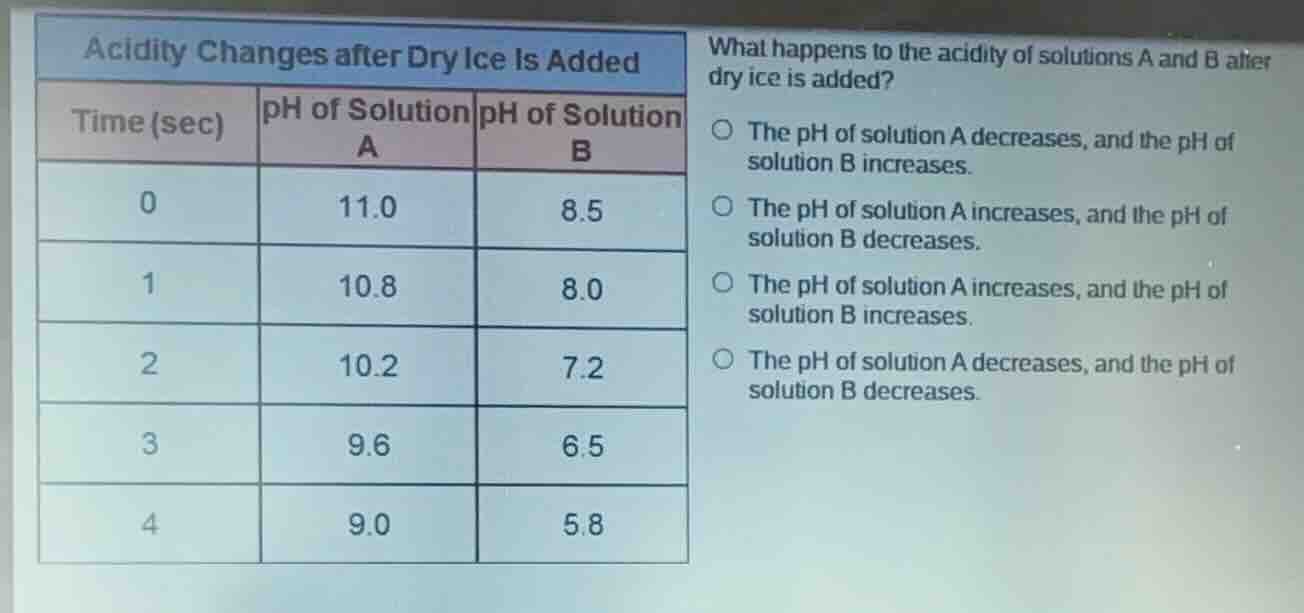
acidity changes after dry ice is added
time (sec) ph of solution a ph of solution b
0 11.0 8.5
1 10.8 8.0
2 10.2 7.2
3 9.6 6.5
4 9.0 5.8
what happens to the acidity of solutions a and b after dry ice is added?
○ the ph of solution a decreases, and the ph of solution b increases.
○ the ph of solution a increases, and the ph of solution b decreases.
○ the ph of solution a increases, and the ph of solution b increases.
○ the ph of solution a decreases, and the ph of solution b decreases.
- pH and acidity have an inverse relationship: lower pH means higher acidity, higher pH means lower acidity.
- From the table, Solution A's pH drops from 11.0 to 9.0 over time as dry ice is added, so its acidity increases (pH decreases).
- Solution B's pH drops from 8.5 to 5.8 over time as dry ice is added, so its acidity also increases (pH decreases).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The pH of solution A decreases, and the pH of solution B decreases.