QUESTION IMAGE
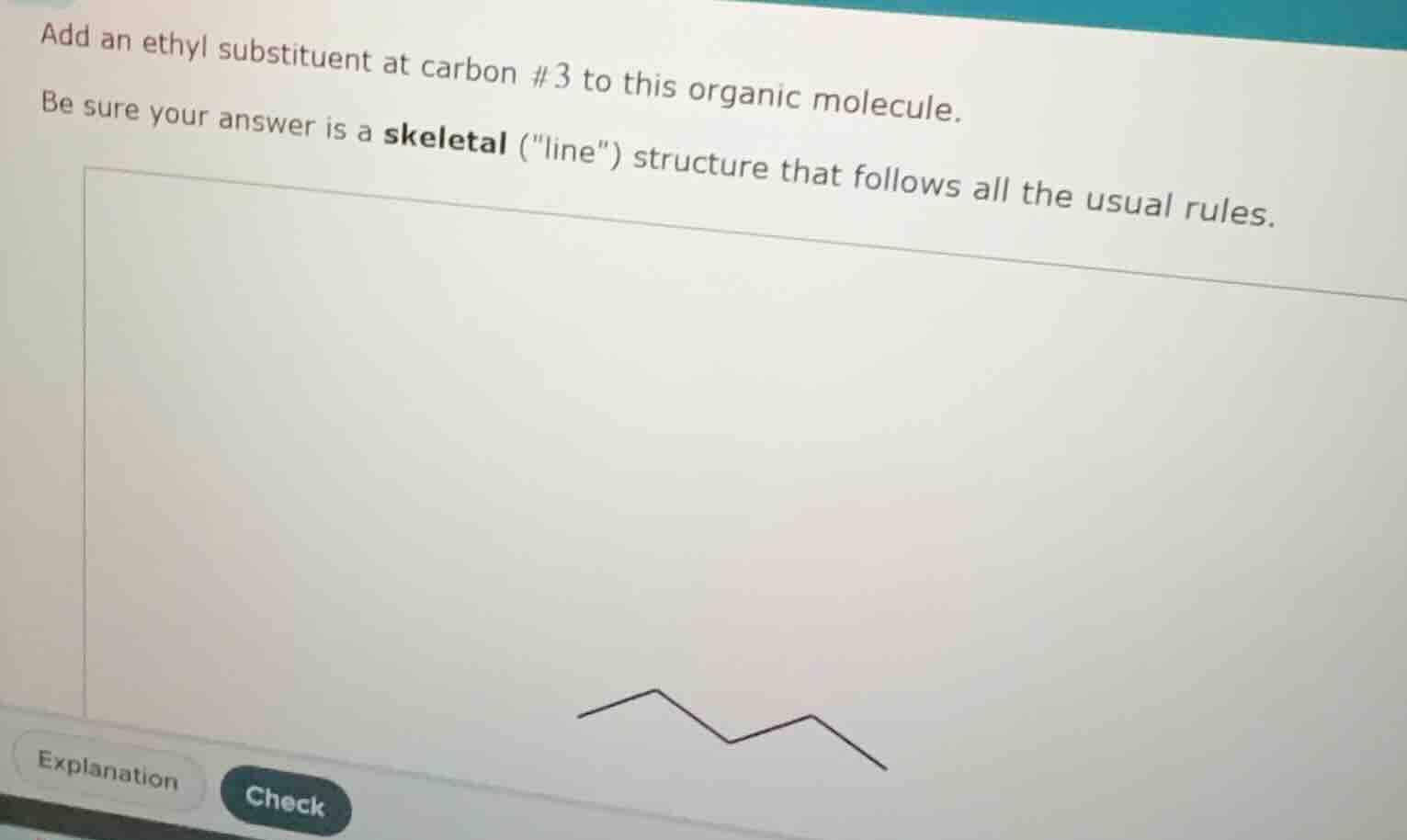
Question
add an ethyl substituent at carbon #3 to this organic molecule. be sure your answer is a skeletal (\line\) structure that follows all the usual rules.
Step1: Identify the parent chain
The given skeletal structure is a straight - chain alkane (assuming it's a simple alkane chain for the base structure). Let's number the carbons from left to right. The first carbon is the left - most end, the second is the next, the third is the middle carbon of the "zig - zag" (since the chain has 4 carbons? Wait, no, looking at the skeletal structure: the first line segment (left - most) is carbon 1 - 2, then 2 - 3, then 3 - 4, then 4 - 5? Wait, no, the skeletal structure as drawn: let's count the vertices (carbons). The left - most start, then a peak (carbon 2), then a valley (carbon 3), then a peak (carbon 4), and the end. Wait, actually, in skeletal structure, each "corner" and each end is a carbon. So the base chain has 4 carbons? Wait, no, the drawing: from left to right, the first line (horizontal? No, it's a zig - zag). Let's number them: carbon 1 (left end), carbon 2 (first peak), carbon 3 (valley), carbon 4 (right peak, end). Wait, no, maybe it's a 4 - carbon chain? Wait, no, the user says "carbon #3", so we need to add an ethyl group ($-CH_2CH_3$) to carbon 3.
Step2: Add the ethyl substituent
The ethyl group has two carbons ($-CH_2 - CH_3$). So at carbon 3 of the parent chain, we attach the ethyl group. The parent chain (let's assume it's a butane? Wait, no, the skeletal structure as drawn: let's represent the parent chain as a 4 - carbon chain (C1 - C2 - C3 - C4). Then at C3, we add $-CH_2CH_3$.
The skeletal structure of the parent chain (before substitution) is:
C2
/
C1 - C3 - C4
\
(wait, no, the original drawing is a zig - zag: C1 (left end) - C2 (up) - C3 (down) - C4 (right end). So numbering: C1 (left), C2 (first corner), C3 (second corner), C4 (right end).
Now, to add ethyl at C3: we draw a line from C3 to a new carbon (C3a), and then from C3a to another carbon (C3b), representing the ethyl group ($-CH_2 - CH_3$).
So the new skeletal structure will be:
C1 - C2 - C3 - C4
|
C3a - C3b
Where C3a is attached to C3, and C3b is attached to C3a. In skeletal terms, this is:
C2
/
C1 - C3 - C4
|
C5 - C6
(where C5 is C3a and C6 is C3b, and the ethyl group is $-CH_2CH_3$ attached to C3)
In proper skeletal drawing, it would look like:
C5
/
C1 - C3 - C4
/
C2
Wait, no, better to draw the parent chain as:
C1 (left) - C2 (up) - C3 (down) - C4 (right)
Then at C3 (the down - corner), we add a branch: from C3, draw a line up (or down, but ethyl is $-CH_2CH_3$, so two carbons) to C5, then from C5 to C6 (right or left). So the skeletal structure becomes:
C5
/
C1 - C3 - C4
/
C2
\
C6? No, ethyl is $-CH_2CH_3$, so the branch from C3 is $-CH_2 - CH_3$, so the first carbon of the ethyl (attached to C3) is $CH_2$ (C5), and the second is $CH_3$ (C6). So the skeletal structure is:
C2
/
C1 - C3 - C4
|
C5 - C6
Which in skeletal (line) structure is:
──C6
/
C1─C3─C4
|
C2
Wait, no, let's do it properly. The parent chain is a 4 - carbon chain (butane? Wait, no, 4 carbons: C1 - C2 - C3 - C4 (zig - zag: C1 left, C2 up, C3 down, C4 right). Adding ethyl at C3: the ethyl group is $-CH_2CH_3$, so we attach a $-CH_2 - CH_3$ to C3. So the skeletal structure is:
CH3
/
C1─C3─C4
|
C2
/
CH2
Wait, no, in skeletal structure, we don't write H or C, just lines. So the ethyl group is two lines (for the two carbons) attached to C3. So the structure is:
C2
/
C1 - C3 - C4
\
C5 - C6
Where C5 - C6 is the ethyl group (C5 is attached to C3, C6 is attached to C…Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The skeletal structure with an ethyl substituent at carbon #3 is:
──
/
─C─C─C─C
|
─
\
──(or in a more standard drawing:
CH₃
/
C₁─C₃─C₄
|
C₂
\
CH₂
But in pure skeletal (line) structure, it's:
──
/
─C─C─C─C
|
─
\
──
)
(Note: The actual correct skeletal structure, assuming the parent chain is a 4 - carbon chain (butane), adding ethyl at carbon 3 (so the parent chain is butane, and the compound is 3 - ethylbutane? Wait, no, 3 - ethylbutane is incorrect, it should be 3 - ethylpentane? Wait, no, maybe the parent chain is 5 carbons? Wait, I think I made a mistake in counting the parent chain. Let's re - examine: the original skeletal structure: the left - most end, then a peak, then a valley, then a peak, and the end. So that's 5 carbons? Wait, no, each "line" segment is a bond between carbons. So the number of carbons is equal to the number of vertices plus 1? No, in skeletal structure, each vertex (corner) and each end is a carbon. So if the structure is drawn as:
Start (C1) - up to C2 - down to C3 - up to C4 - end (C5). So that's a 5 - carbon chain (pentane). Then carbon #3 is the middle carbon (C3). Then adding ethyl at C3: the ethyl group ($-CH_2CH_3$) attached to C3.
So the parent chain is pentane (C1 - C2 - C3 - C4 - C5). Then at C3, we add $-CH_2CH_3$.
The skeletal structure of pentane is:
C2
/
C1 - C3 - C5
\
C4(Wait, no, pentane's skeletal structure is a zig - zag with 5 carbons: C1 (left) - C2 (up) - C3 (down) - C4 (up) - C5 (right). Then at C3, we add the ethyl group.
So the correct skeletal structure with ethyl at C3 (of pentane) is:
C2
/
C1 - C3 - C5
|
C3a - C3b
\
C4Where C3a - C3b is the ethyl group ($-CH_2CH_3$). In line structure, this is:
C3b
/
C1─C3─C5
|
C3a
\
C4
/
C2
Wait, no, to draw it properly, the pentane chain is:
C1 (left) - C2 (up) - C3 (down) - C4 (up) - C5 (right)
At C3 (the down - facing carbon), we attach a $-CH_2CH_3$ group. So we draw a line from C3 to C6 (first carbon of ethyl), then from C6 to C7 (second carbon of ethyl). So the skeletal structure is:
C7
/
C1─C3─C5
|
C6
\
C4
/
C2
In line (skeletal) notation, this is:
──
/
─C─C─C─C─C
|
──
\
─
/
─
(But this is getting confusing. The key is that the ethyl group has two carbons, and we attach it to carbon #3 of the parent chain, drawing the skeletal structure with lines representing bonds, and carbons at the corners and ends.)
The correct skeletal structure (assuming the parent chain is a 5 - carbon chain, pentane, and we add ethyl at carbon 3, resulting in 3 - ethylpentane) is:
──
/
─C─C─C─C─C
|
──
\
─(where the middle C (third from left) has a two - carbon branch (ethyl) attached)