QUESTION IMAGE
Question
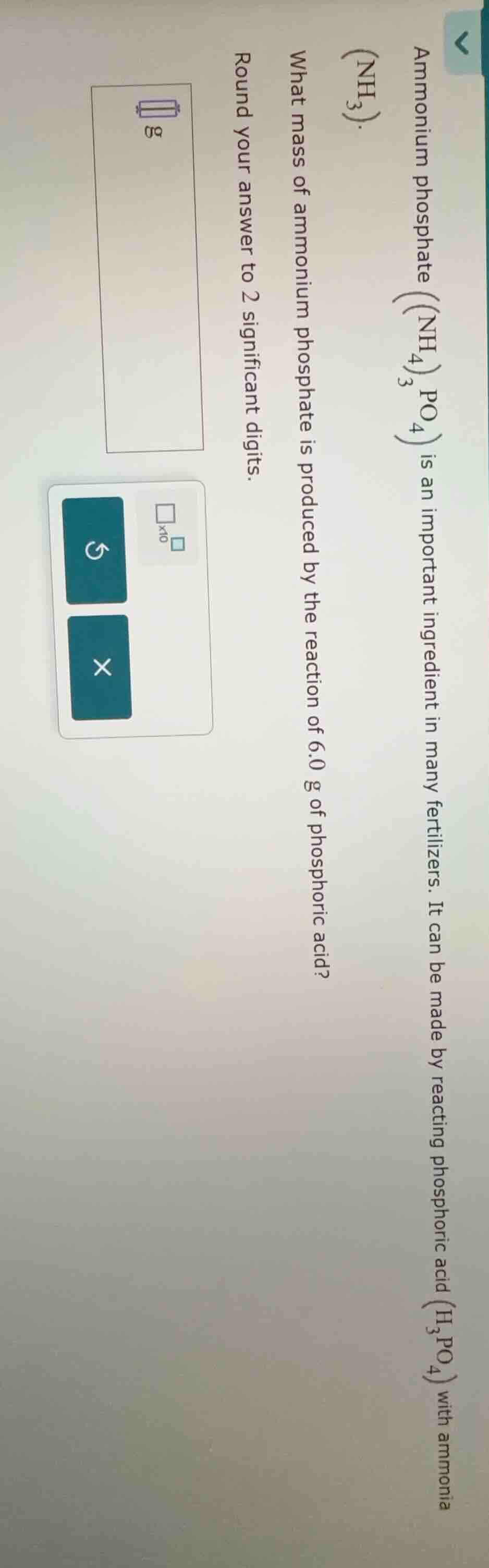
ammonium phosphate $\ce{(nh_{4})_{3}po_{4}}$ is an important ingredient in many fertilizers. it can be made by reacting phosphoric acid $\ce{(h_{3}po_{4})}$ with ammonia $\ce{(nh_{3})}$. what mass of ammonium phosphate is produced by the reaction of 6.0 g of phosphoric acid? round your answer to 2 significant digits.
Step1: Write the balanced reaction
The reaction between phosphoric acid ($\ce{H3PO4}$) and ammonia ($\ce{NH3}$) to form ammonium phosphate ($\ce{(NH4)3PO4}$) is:
$$\ce{H3PO4 + 3NH3 -> (NH4)3PO4}$$
Step2: Calculate moles of $\ce{H3PO4}$
Molar mass of $\ce{H3PO4}$:
$M(\ce{H3PO4}) = 3(1.008) + 30.97 + 4(16.00) = 97.994\ \text{g/mol}$
Moles of $\ce{H3PO4}$:
$n(\ce{H3PO4}) = \frac{\text{mass}}{\text{molar mass}} = \frac{6.0\ \text{g}}{97.994\ \text{g/mol}} \approx 0.0612\ \text{mol}$
Step3: Relate moles of $\ce{H3PO4}$ to $\ce{(NH4)3PO4}$
From the balanced equation, $1\ \text{mol}\ \ce{H3PO4}$ produces $1\ \text{mol}\ \ce{(NH4)3PO4}$.
Thus, $n(\ce{(NH4)3PO4}) = n(\ce{H3PO4}) \approx 0.0612\ \text{mol}$
Step4: Calculate molar mass of $\ce{(NH4)3PO4}$
Molar mass of $\ce{(NH4)3PO4}$:
$M(\ce{(NH4)3PO4}) = 3(14.01 + 4(1.008)) + 30.97 + 4(16.00)$
$= 3(18.042) + 30.97 + 64.00$
$= 54.126 + 30.97 + 64.00 = 149.096\ \text{g/mol}$
Step5: Calculate mass of $\ce{(NH4)3PO4}$
Mass of $\ce{(NH4)3PO4}$:
$m = n \times M = 0.0612\ \text{mol} \times 149.096\ \text{g/mol} \approx 9.1\ \text{g}$ (rounded to 2 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{9.1\ \text{g}}$