QUESTION IMAGE
Question
analyze a chemical reaction
- the equation that represents
the chemical reaction shown
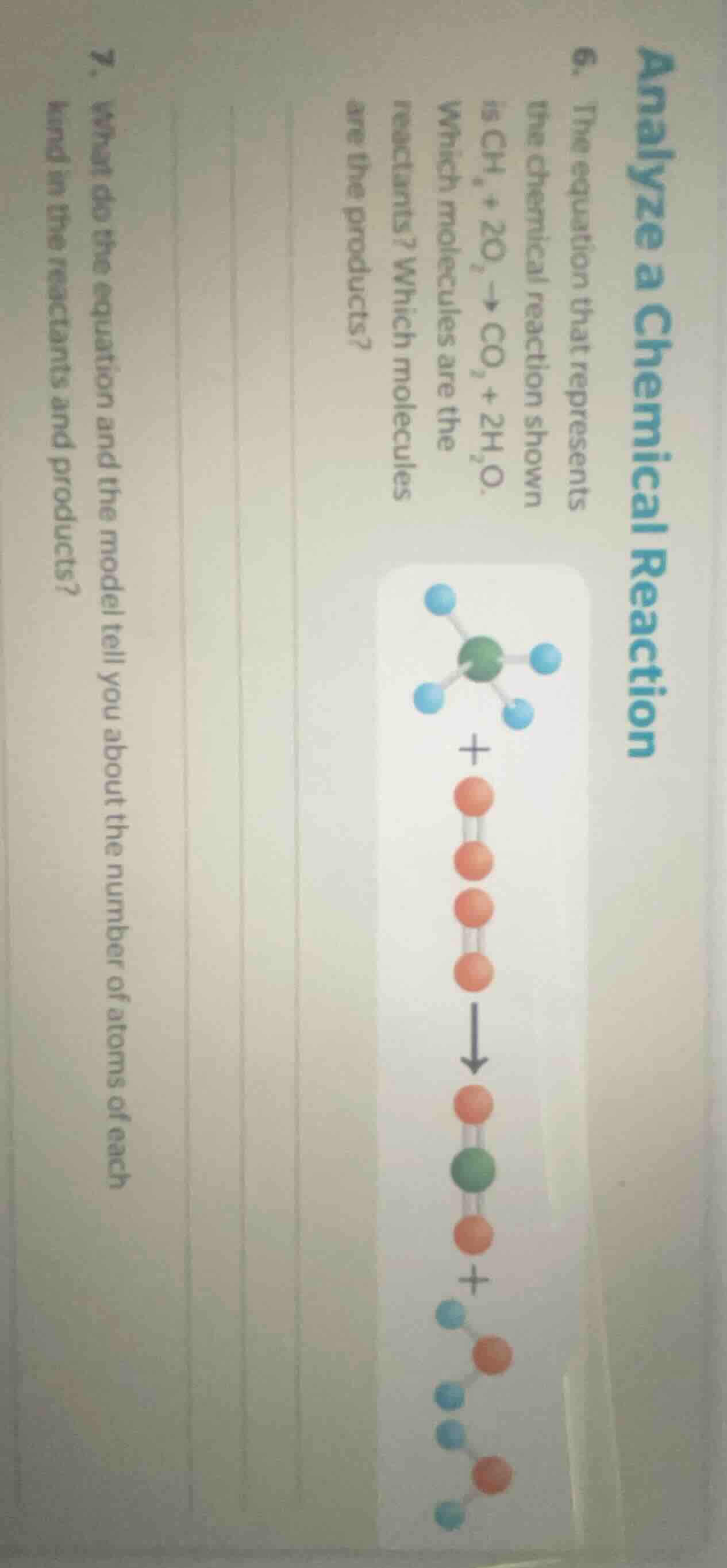
is $\ce{ch_{4} + 2o_{2} -> co_{2} + 2h_{2}o}$.
which molecules are the
reactants? which molecules
are the products?
(there is a molecular model diagram showing $\ce{ch_{4}}$ + $\ce{o_{2}}$ -> $\ce{co_{2}}$ + $\ce{h_{2}o}$)
- what do the equation and the model tell you about the number of atoms of each
kind in the reactants and products?
Question 6
In a chemical reaction equation, reactants are the substances on the left side of the arrow (the starting materials), and products are on the right side (the substances formed). The equation given is $\ce{CH_{4} + 2O_{2} -> CO_{2} + 2H_{2}O}$. So we identify the molecules on each side.
To determine the number of atoms of each kind in reactants and products, we analyze the chemical formula of each compound and the coefficients. For reactants: $\ce{CH_{4}}$ has 1 C and 4 H; $\ce{2O_{2}}$ has 4 O (since 2 molecules of $\ce{O_{2}}$: 2×2 = 4 O). For products: $\ce{CO_{2}}$ has 1 C and 2 O; $\ce{2H_{2}O}$ has 4 H (2×2) and 2 O (2×1), so total O in products is 2 + 2 = 4. So we count atoms for each element (C, H, O) in reactants and products.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Reactants (molecules): Methane ($\ce{CH_{4}}$) and oxygen ($\ce{O_{2}}$)
- Products (molecules): Carbon dioxide ($\ce{CO_{2}}$) and water ($\ce{H_{2}O}$)