QUESTION IMAGE
Question
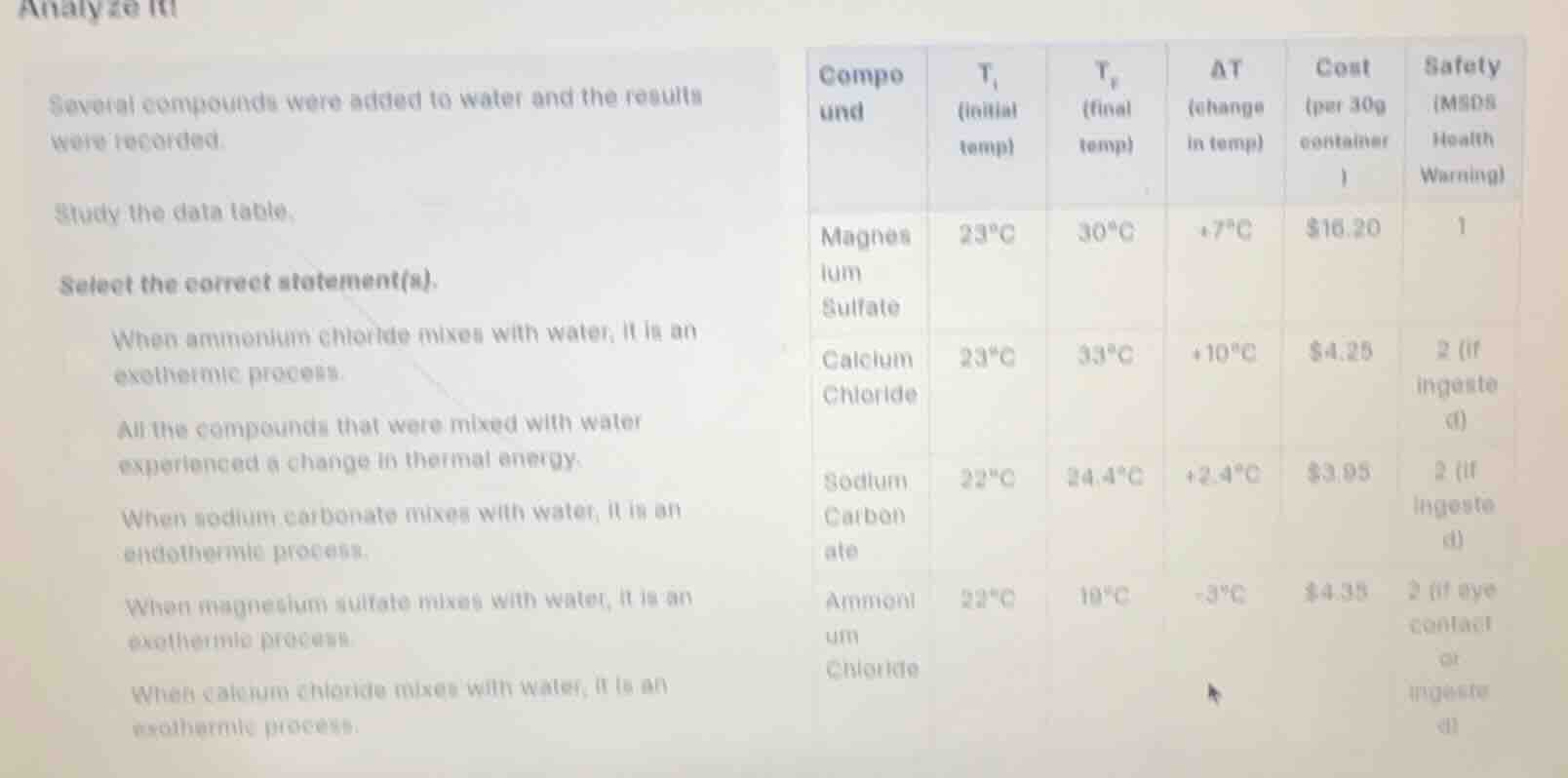
analyze it!several compounds were added to water and the results were recorded.study the data table.select the correct statement(s).when ammonium chloride mixes with water, it is an exothermic process.all the compounds that were mixed with water experienced a change in thermal energy.when sodium carbonate mixes with water, it is an endothermic process.when magnesium sulfate mixes with water, it is an exothermic process.when calcium chloride mixes with water, it is an exothermic process.
Brief Explanations
- For ammonium chloride: $\Delta T = 19^\circ\text{C} - 22^\circ\text{C} = -3^\circ\text{C}$. A temperature decrease means energy is absorbed from water, so this is an endothermic process, not exothermic.
- Every compound has a non-zero $\Delta T$, meaning all experienced a thermal energy change (either releasing or absorbing heat).
- Sodium carbonate has $\Delta T = 24.4^\circ\text{C} - 22^\circ\text{C} = +2.4^\circ\text{C}$. A temperature increase means energy is released into water, so this is an exothermic process, not endothermic.
- Magnesium sulfate has $\Delta T = 30^\circ\text{C} - 23^\circ\text{C} = +7^\circ\text{C}$. A temperature increase means energy is released into water, so this is an exothermic process.
- Calcium chloride has $\Delta T = 33^\circ\text{C} - 23^\circ\text{C} = +10^\circ\text{C}$. A temperature increase means energy is released into water, so this is an exothermic process.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- All the compounds that were mixed with water experienced a change in thermal energy.
- When magnesium sulfate mixes with water, it is an exothermic process.
- When calcium chloride mixes with water, it is an exothermic process.