QUESTION IMAGE
Question
anthocyanin experiment
determining concentration
anthocyanins are red or purple-colored molecules
found in grapes, tart cherries, and red kidney
beans, among other foods.
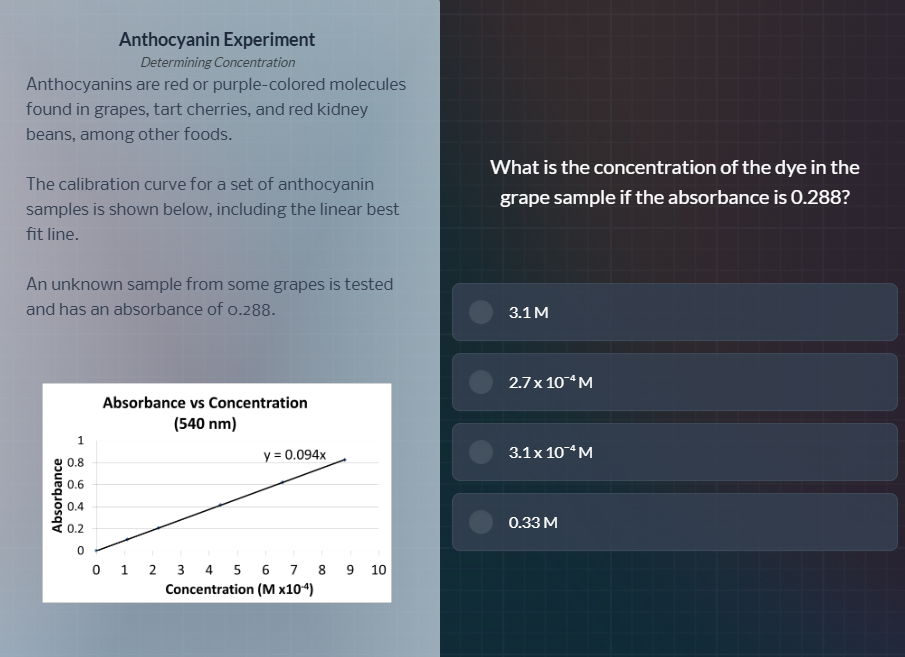
the calibration curve for a set of anthocyanin
samples is shown below, including the linear best
fit line.
an unknown sample from some grapes is tested
and has an absorbance of 0.288.
absorbance vs concentration
(540 nm)
y = 0.094x
what is the concentration of the dye in the
grape sample if the absorbance is 0.288?
3.1 m
2.7 x 10⁻⁴ m
3.1 x 10⁻⁴ m
0.33 m
Step1: Identify variables from the equation
The linear fit equation is $y = 0.094x$, where $y$ = absorbance, $x$ = concentration ($\text{M} \times 10^{-4}$).
Step2: Substitute absorbance value
Substitute $y = 0.288$ into the equation:
$0.288 = 0.094x$
Step3: Solve for x (concentration)
Rearrange to solve for $x$:
$x = \frac{0.288}{0.094} \approx 3.06$
Since $x$ is in $\text{M} \times 10^{-4}$, the concentration is $3.1 \times 10^{-4}\ \text{M}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $3.1 \times 10^{-4}$ M