QUESTION IMAGE
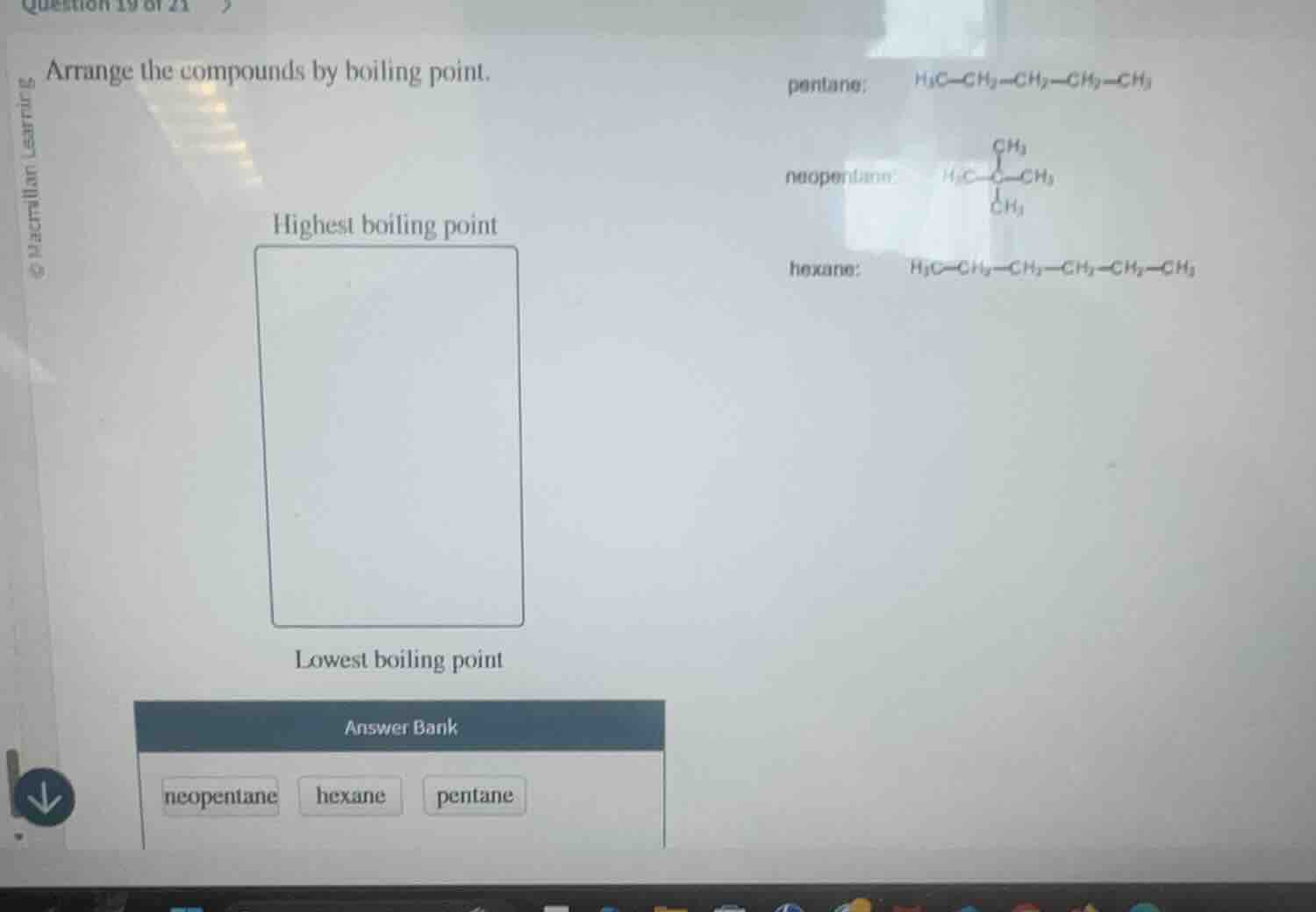
Question
arrange the compounds by boiling point.
highest boiling point
lowest boiling point
answer bank
neopentane hexane pentane
pentane: $\ce{h3c-ch2-ch2-ch2-ch3}$
neopentane: $\ce{h3c-\overset{ch3}{\underset{ch3}{c}}-ch3}$
hexane: $\ce{h3c-ch2-ch2-ch2-ch2-ch3}$
Boiling points of alkanes depend on molecular weight (higher molecular weight = stronger London dispersion forces = higher boiling point) and branching (more branching = less surface area = weaker London dispersion forces = lower boiling point). Hexane has 6 carbons (highest molecular weight), pentane has 5 carbons (linear, no branching), neopentane has 5 carbons but is highly branched.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Highest boiling point: hexane ($\text{H}_3\text{C-CH}_2\text{-CH}_2\text{-CH}_2\text{-CH}_2\text{-CH}_3$)
pentane ($\text{H}_3\text{C-CH}_2\text{-CH}_2\text{-CH}_2\text{-CH}_3$)
Lowest boiling point: neopentane ($\text{H}_3\text{C-C(CH}_3\text{)}_3\text{-CH}_3$)