QUESTION IMAGE
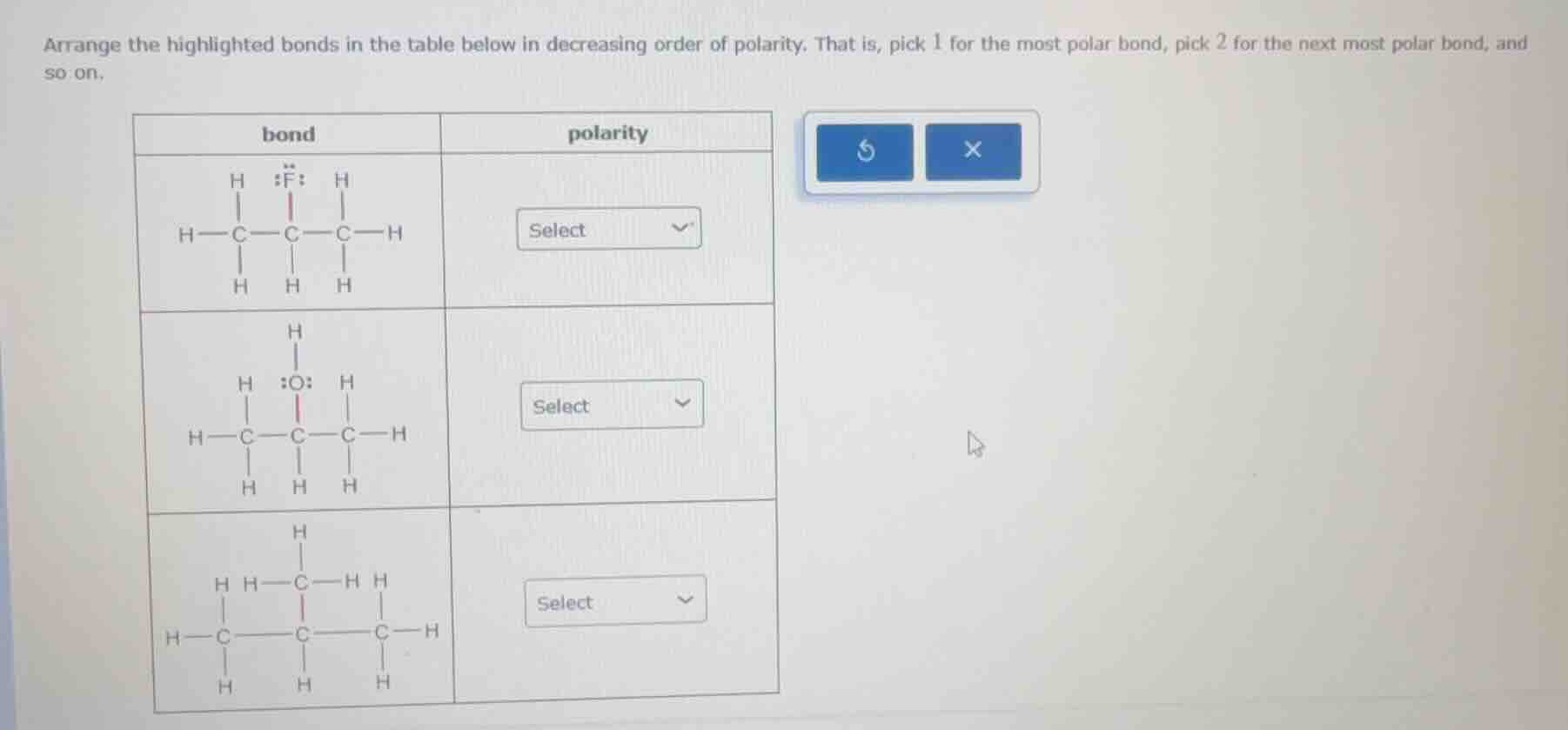
Question
arrange the highlighted bonds in the table below in decreasing order of polarity. that is, pick 1 for the most polar bond, pick 2 for the next most polar bond, and so on.
bond polarity
select
select
select
To determine the polarity of bonds, we use the concept of electronegativity difference between the bonded atoms. The greater the electronegativity difference, the more polar the bond.
Step 1: Identify the bonds
- First bond: \( \text{C-F} \) (carbon - fluorine)
- Second bond: \( \text{C-O} \) (carbon - oxygen)
- Third bond: \( \text{C-C} \) (carbon - carbon)
Step 2: Recall electronegativity values
- Electronegativity of \( \text{C} \): ~2.5
- Electronegativity of \( \text{F} \): ~4.0
- Electronegativity of \( \text{O} \): ~3.5
- Electronegativity of \( \text{C} \): ~2.5
Step 3: Calculate electronegativity differences
- For \( \text{C-F} \): \( |4.0 - 2.5| = 1.5 \)
- For \( \text{C-O} \): \( |3.5 - 2.5| = 1.0 \)
- For \( \text{C-C} \): \( |2.5 - 2.5| = 0.0 \)
Step 4: Rank by polarity
The greater the electronegativity difference, the more polar the bond. So the order from most polar to least polar is: \( \text{C-F} \) (1), \( \text{C-O} \) (2), \( \text{C-C} \) (3)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- For the \( \text{C-F} \) bond (first row): 1
- For the \( \text{C-O} \) bond (second row): 2
- For the \( \text{C-C} \) bond (third row): 3