QUESTION IMAGE
Question
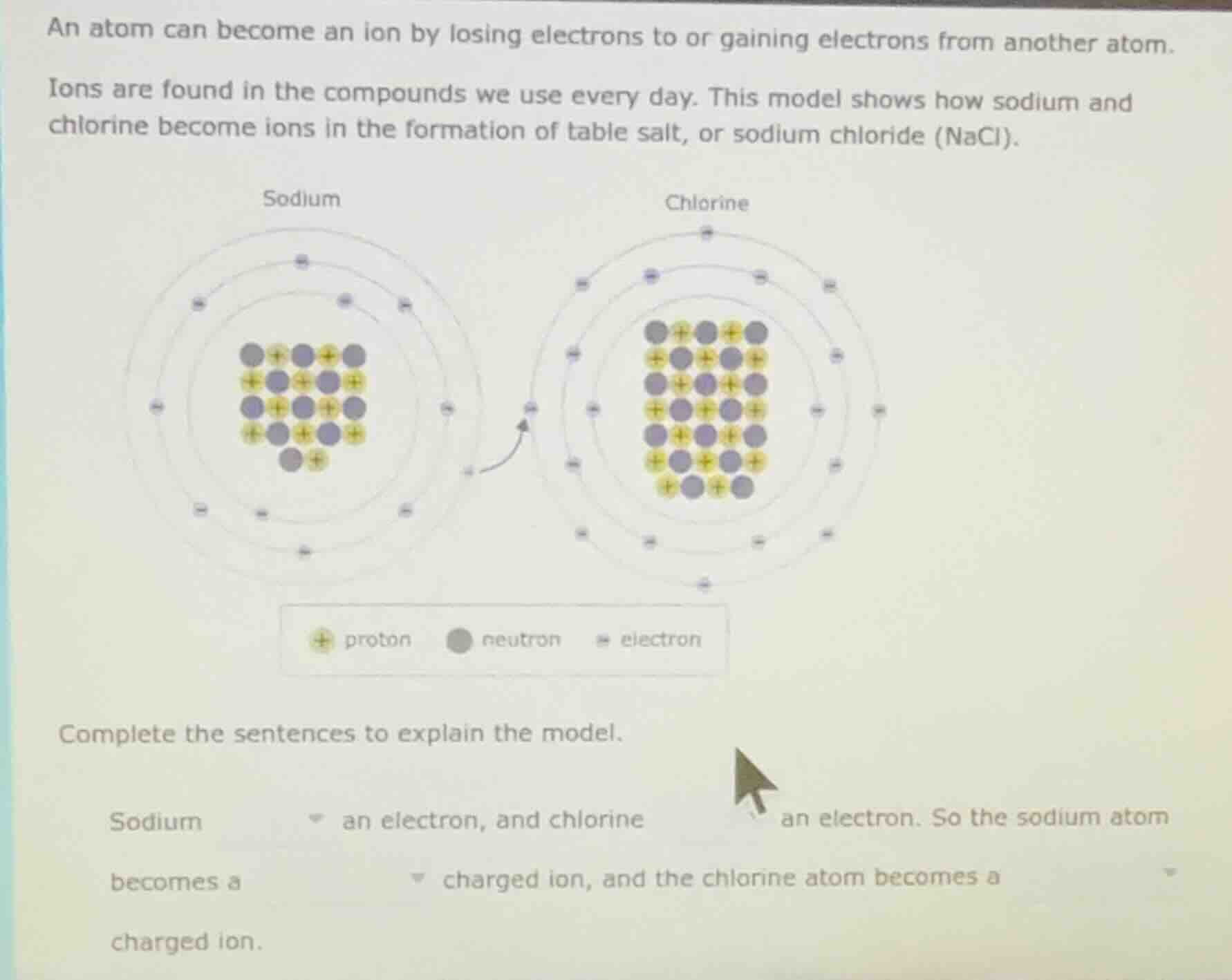
an atom can become an ion by losing electrons to or gaining electrons from another atom. ions are found in the compounds we use every day. this model shows how sodium and chlorine become ions in the formation of table salt, or sodium chloride (nacl). image of sodium and chlorine atom models with electron transfer complete the sentences to explain the model. sodium dropdown an electron, and chlorine dropdown an electron. so the sodium atom becomes a dropdown charged ion, and the chlorine atom becomes a dropdown charged ion.
- From the model (and chemical knowledge), sodium has one valence electron and tends to lose it to achieve a stable electron configuration. Chlorine has 7 valence electrons and tends to gain one electron to complete its octet.
- When an atom loses an electron, it has more protons than electrons, so it becomes positively charged (a cation). When an atom gains an electron, it has more electrons than protons, so it becomes negatively charged (an anion).
So, sodium loses an electron, chlorine gains an electron. Sodium becomes a positively charged ion, chlorine becomes a negatively charged ion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Sodium \(\boldsymbol{\text{loses}}\) an electron, and chlorine \(\boldsymbol{\text{gains}}\) an electron. So the sodium atom becomes a \(\boldsymbol{\text{positively}}\) charged ion, and the chlorine atom becomes a \(\boldsymbol{\text{negatively}}\) charged ion.