QUESTION IMAGE
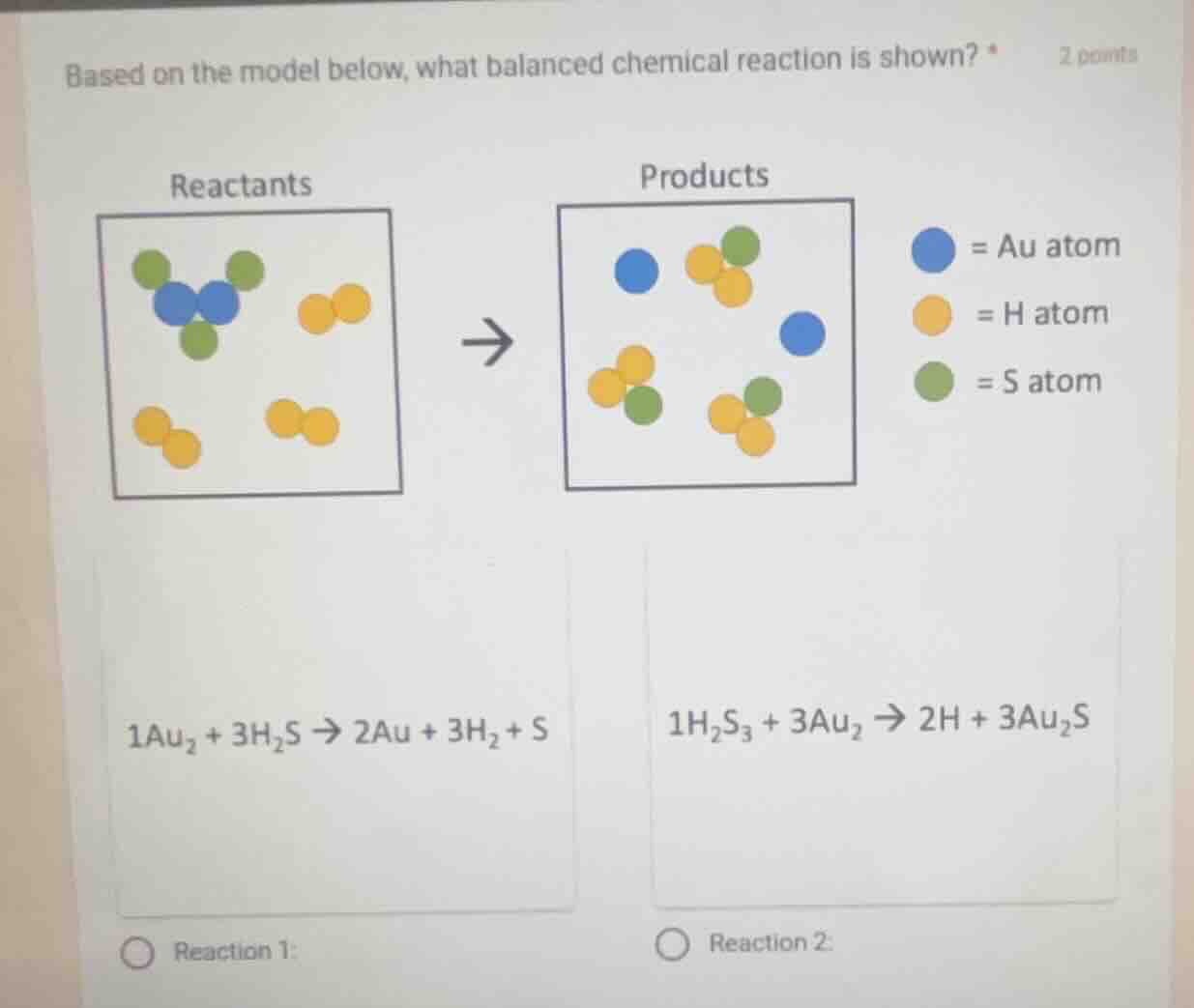
Question
based on the model below, what balanced chemical reaction is shown? *
reactants
products
= au atom
= h atom
= s atom
1au₂ + 3h₂s → 2au + 3h₂ + s
1h₂s₃ + 3au₂ → 2h + 3au₂s
reaction 1:
reaction 2:
Step1: Count reactant atoms
- Au: 2 (from 1 $\text{Au}_2$ molecule)
- H: $3 \times 2 = 6$ (from 3 $\text{H}_2$ molecules)
- S: 3 (from 3 $\text{H}_2\text{S}$ units, 1 S each)
Total reactant atoms: 2 Au, 6 H, 3 S
Step2: Count product atoms
- Au: 2 (2 single Au atoms)
- H: $3 \times 2 = 6$ (from 3 $\text{H}_2$ molecules)
- S: 3 (1 free S + 2 S in $\text{H}_2\text{S}$-like product units)
Total product atoms: 2 Au, 6 H, 3 S
Step3: Match to reactions
Reaction 1 has equal atom counts on both sides, matching the model. Reaction 2 has mismatched atom counts (e.g., 6 Au on reactants, 6 Au on products but H and S counts do not match the model).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reaction 1: $1\text{Au}_2 + 3\text{H}_2\text{S}
ightarrow 2\text{Au} + 3\text{H}_2 + \text{S}$