QUESTION IMAGE
Question
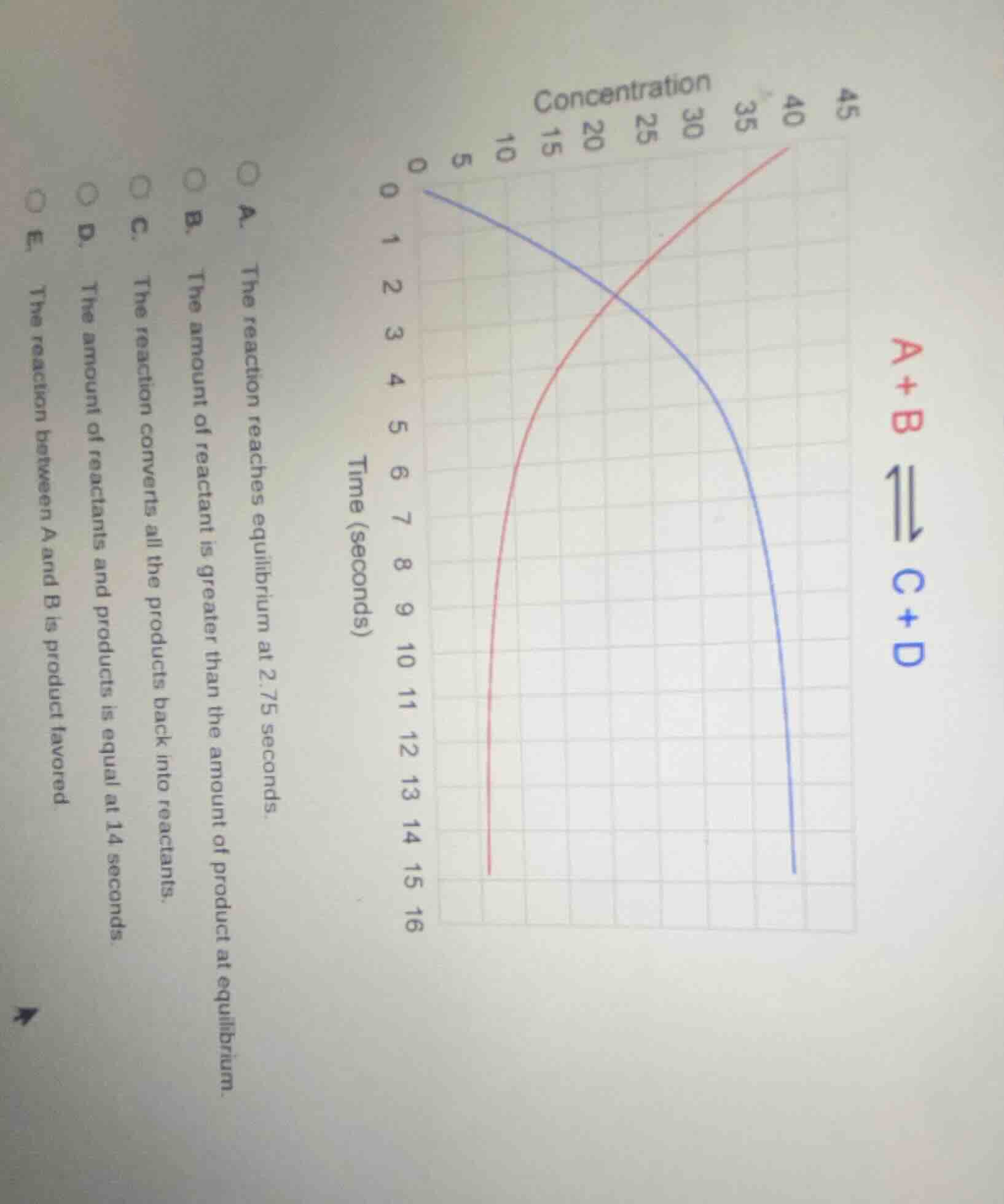
a+bc+dtime (seconds)concentrationa. the reaction reaches equilibrium at 2.75 seconds.b. the amount of reactant is greater than the amount of product at equilibrium.c. the reaction converts all the products back into reactants.d. the amount of reactants and products is equal at 14 seconds.e. the reaction between a and b is product favored.
To solve this, we analyze each option:
- Option A: A reaction reaches equilibrium when concentrations stop changing. At 2.75 seconds, concentrations are still changing (curves are sloping), so A is wrong.
- Option B: At equilibrium (when curves flatten), the blue curve (reactants, A+B) and red curve (products, C+D) – the reactant concentration (blue) is lower than product (red) at equilibrium? Wait, no, wait: Wait, the red curve is A+B? Wait, no, the legend: red is A+B, blue is C+D. Wait, initial concentration of A+B (red) is 0? No, wait, at time 0, blue (C+D) is 0? Wait, no, the x-axis is concentration, y-axis is time. Wait, no, the y-axis is time (seconds), x-axis is concentration. Wait, at time 0, blue (C+D) has concentration 0? No, at time 0, blue (C+D) starts at 0? Wait, no, the graph: the blue curve starts at (0,0) time? No, time is y-axis (0 to 16 seconds), concentration x-axis (0 to 45). So at time 0 (y=0), blue (C+D) has concentration 0? No, wait, the blue curve starts at (0,0) in concentration? No, at time 0 (y=0), the blue curve (C+D) is at concentration 0? Wait, no, the red curve (A+B) starts at concentration 0? No, this is confusing. Wait, the reaction is A + B ⇌ C + D. So initially (time 0), A+B (reactants) should be high, C+D (products) low. Wait, but the red curve (A+B) starts at concentration 0? No, maybe the axes are reversed? Wait, no, the x-axis is "Concentration", y-axis is "Time (seconds)". So each point (x, y) is concentration x at time y. So at time 0 (y=0), blue (C+D) is at x=0 (concentration 0), red (A+B) is at x=0? No, that can't be. Wait, maybe the blue curve is C+D (products) and red is A+B (reactants). Wait, as time increases (y increases), red (A+B) concentration increases? No, that doesn't make sense. Wait, no – in a reaction A + B → C + D, reactants (A+B) should decrease, products (C+D) increase over time until equilibrium. So the curve for A+B (reactants) should decrease, C+D (products) increase. But in the graph, red (A+B) is increasing, blue (C+D) is increasing? No, that's not possible. Wait, maybe the red curve is C+D (products) and blue is A+B (reactants). Wait, let's re-express:
- If reaction is A + B ⇌ C + D, then:
- Reactants (A+B): concentration decreases over time until equilibrium.
- Products (C+D): concentration increases over time until equilibrium.
But in the graph, red and blue are both increasing? That can't be. Wait, maybe the axes are swapped: x-axis is time, y-axis is concentration. That would make more sense. Maybe the graph is misread. Let's assume x-axis is time (seconds), y-axis is concentration. Then:
- At time 0, A+B (red) has high concentration, C+D (blue) has 0.
- As time increases, A+B (red) decreases, C+D (blue) increases, until they flatten (equilibrium).
But the given graph has x-axis as concentration, y-axis as time. So maybe it's a plot of concentration vs. time, with x=concentration, y=time. So each curve is concentration over time: for A+B (red), as time (y) increases, concentration (x) increases? No, that would mean A+B is being produced, which is opposite. This is confusing. Wait, maybe the correct approach is to look at equilibrium: equilibrium is when concentrations stop changing (curves become horizontal). Looking at the graph, the curves start to flatten around, say, time 10-15 seconds? Wait, no, the blue curve (C+D) is still increasing at time 15, red (A+B) too? No, that can't be. Wait, maybe the question's options: let's re-express the options.
Wait, the options:
B: "The amount of reactant is greater than the amount of product at equilibrium."
Wa…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
E. The reaction between A and B is product favored