QUESTION IMAGE
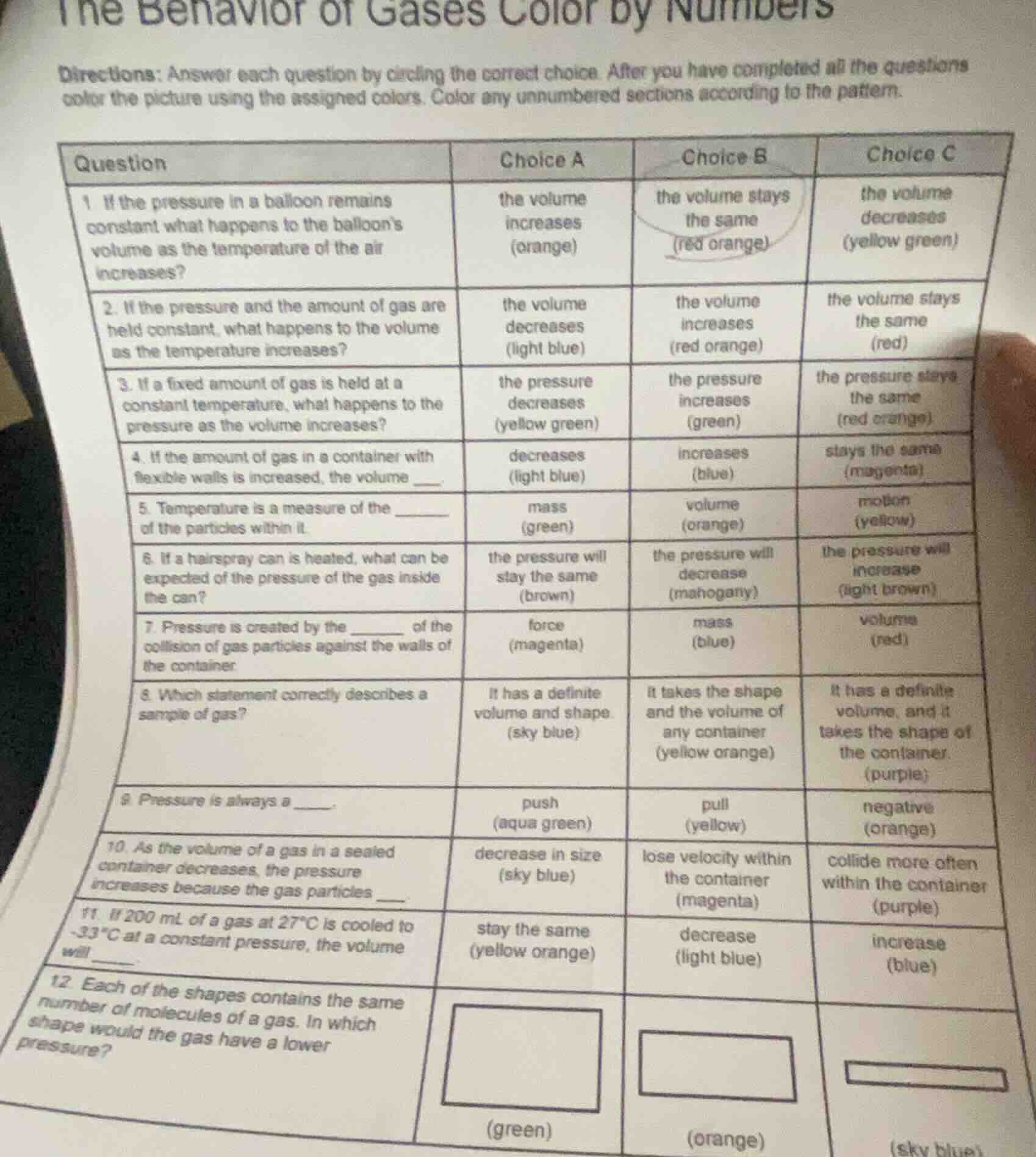
Question
the behavior of gases color by numbers
directions: answer each question by circling the correct choice. after you have completed all the questions color the picture using the assigned colors. color any unnumbered sections according to the pattern.
| question | choice a | choice b | choice c |
|---|---|---|---|
| 2. if the pressure and the amount of gas are held constant, what happens to the volume as the temperature increases? | the volume decreases (light blue) | the volume increases (red orange) | the volume stays the same (red) |
| 3. if a fixed amount of gas is held at a constant temperature, what happens to the pressure as the volume increases? | the pressure decreases (yellow green) | the pressure increases (green) | the pressure stays the same (red orange) |
| 4. if the amount of gas in a container with flexible walls is increased, the volume _____. | decreases (light blue) | increases (blue) | stays the same (magenta) |
| 5. temperature is a measure of the ______ of the particles within it. | mass (green) | volume (orange) | motion (yellow) |
| 6. if a hairspray can is heated, what can be expected of the pressure of the gas inside the can? | the pressure will stay the same (brown) | the pressure will decrease (mahogany) | the pressure will increase (light brown) |
| 7. pressure is created by the ______ of the collision of gas particles against the walls of the container. | force (magenta) | mass (blue) | volume (red) |
| 8. which statement correctly describes a sample of gas? | it has a definite volume and shape (sky blue) | it takes the shape and the volume of any container (yellow orange) | it has a definite volume, and it takes the shape of the container. (purple) |
| 9. pressure is always a ______. | push (aqua green) | pull (yellow) | negative (orange) |
| 10. as the volume of a gas in a sealed container decreases, the pressure increases because the gas particles _____. | decrease in size (sky blue) | lose velocity within the container (magenta) | collide more often within the container (purple) |
| 11. if 200 ml of a gas at 27°c is cooled to -33°c at a constant pressure, the volume will ______. | stay the same (yellow orange) | decrease (light blue) | increase (blue) |
| 12. each of the shapes contains the same number of molecules of a gas. in which shape would the gas have a lower pressure? | (square, green) | (rectangle, orange) | (long narrow rectangle, sky blue) |
Step1: Apply Charles's Law (Q1)
Charles's Law: $\frac{V_1}{T_1}=\frac{V_2}{T_2}$ (constant $P,n$). As $T$ increases, $V$ increases.
Step2: Apply Charles's Law (Q2)
Charles's Law: $\frac{V_1}{T_1}=\frac{V_2}{T_2}$ (constant $P,n$). As $T$ increases, $V$ increases.
Step3: Apply Boyle's Law (Q3)
Boyle's Law: $P_1V_1=P_2V_2$ (constant $T,n$). As $V$ increases, $P$ decreases.
Step4: Relate moles to volume (Q4)
At constant $P,T$, $V\propto n$. As $n$ increases, $V$ increases.
Step5: Define temperature (Q5)
Temperature measures particle kinetic motion.
Step6: Apply Gay-Lussac's Law (Q6)
Gay-Lussac's Law: $\frac{P_1}{T_1}=\frac{P_2}{T_2}$ (constant $V,n$). As $T$ increases, $P$ increases.
Step7: Define gas pressure (Q7)
Pressure comes from particle collision force on walls.
Step8: Identify gas properties (Q8)
Gases take container shape/volume, no fixed form.
Step9: Define pressure (Q9)
Pressure is a pushing force on surfaces.
Step10: Explain pressure-volume relation (Q10)
Smaller $V$ = more frequent particle collisions, higher $P$.
Step11: Calculate volume change (Q11)
Convert $T$ to Kelvin: $T_1=27+273=300\ \text{K}$, $T_2=-33+273=240\ \text{K}$.
Charles's Law: $V_2=V_1\times\frac{T_2}{T_1}=200\times\frac{240}{300}=160\ \text{mL}$. Volume decreases.
Step12: Relate volume to pressure (Q12)
Boyle's Law: $P\propto\frac{1}{V}$ (constant $T,n$). Larger volume = lower pressure.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- A. the volume increases (orange)
- B. the volume increases (red orange)
- A. the pressure decreases (yellow green)
- B. increases (blue)
- C. motion (yellow)
- C. the pressure will increase (light brown)
- A. force (magenta)
- B. it takes the shape and the volume of any container (yellow orange)
- A. push (aqua green)
- C. collide more often within the container (purple)
- B. decrease (light blue)
- C. <long rectangular shape> (sky blue)