QUESTION IMAGE
Question
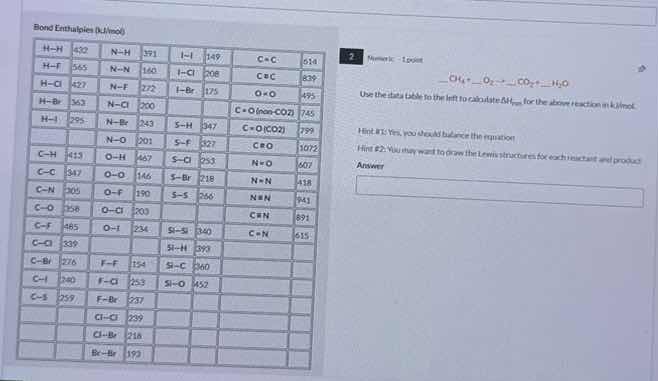
bond enthalpies (kj/mol)
h–h 432; n–h 391; i–i 149; c=c 614
h–f 565; n–n 160; i–cl 208; c≡c 839
h–cl 427; n–cl 200; i–br 175; o=o 495
h–i 299; n–br 243; s–h 347; c=o (gas, co₂) 799? wait, no, maybe c=o (co) 755? wait, the table has: c=o (co₂)? wait, the users table: c=o (co₂)? wait, the ocr: c=o (gas, co₂) 799? wait, no, the original: c–o (co₂) 799? wait, maybe typo. then the reaction: _ch₄ + _o₂ → _co₂ + _h₂o. use the data table to the left to calculate δhᵣₓₙ for the above reaction in kj/mol. hint #1: yes, you should balance the reaction! hint #2: you may want to draw the lewis structures for each reactant and product. answer
Step1: Balance the combustion reaction
The unbalanced reaction is $\text{O}_2\text{H}_4 + \text{O}_2
ightarrow \text{CO}_2 + \text{H}_2\text{O}$. The balanced reaction is:
Step2: Count bonds broken (reactants)
- $\text{C}_2\text{H}_4$: 1 $\text{C=C}$ bond, 4 $\text{C-H}$ bonds
- $3\text{O}_2$: 3 $\text{O=O}$ bonds
Calculate total bond enthalpy of broken bonds:
$$\Delta H_{\text{broken}} = (614 + 4\times413) + 3\times498 = (614 + 1652) + 1494 = 2266 + 1494 = 3760\ \text{kJ/mol}$$
Step3: Count bonds formed (products)
- $2\text{CO}_2$: 4 $\text{C=O}$ bonds (each $\text{CO}_2$ has 2 $\text{C=O}$)
- $2\text{H}_2\text{O}$: 4 $\text{O-H}$ bonds (each $\text{H}_2\text{O}$ has 2 $\text{O-H}$)
Calculate total bond enthalpy of formed bonds:
$$\Delta H_{\text{formed}} = 4\times799 + 4\times467 = 3196 + 1868 = 5064\ \text{kJ/mol}$$
Step4: Calculate reaction enthalpy
Reaction enthalpy is $\Delta H_{\text{rxn}} = \Delta H_{\text{broken}} - \Delta H_{\text{formed}}$
$$\Delta H_{\text{rxn}} = 3760 - 5064 = -1304\ \text{kJ/mol}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{-1304}$ kJ/mol