QUESTION IMAGE
Question
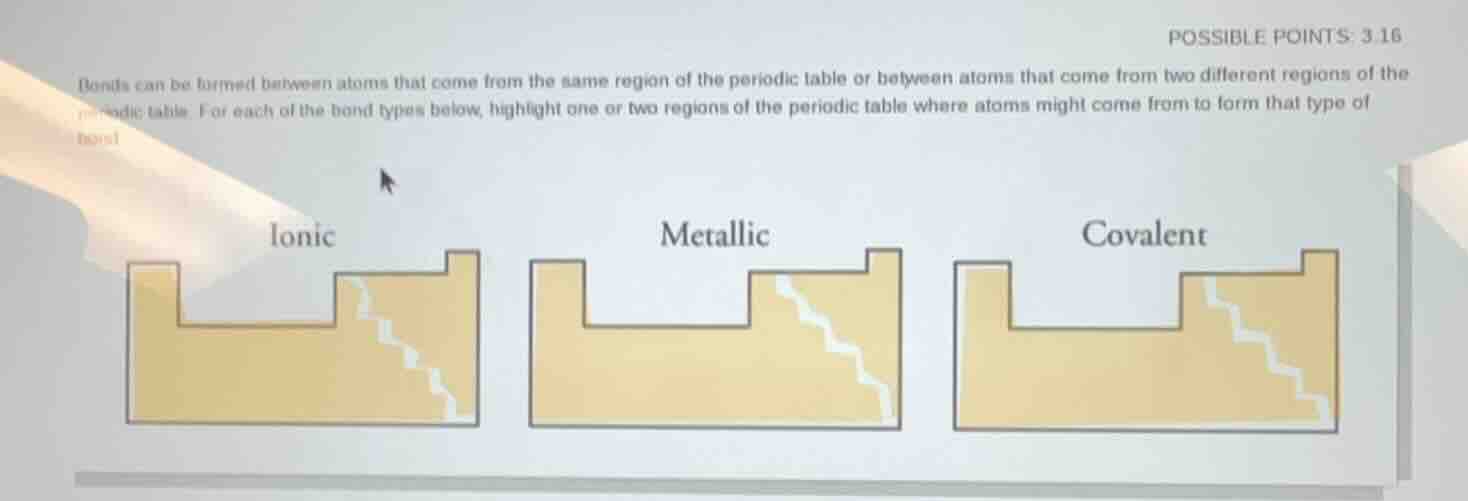
bonds can be formed between atoms that come from the same region of the periodic table or between atoms that come from two different regions of the periodic table. for each of the bond types below, highlight one or two regions of the periodic table where atoms might come from to form that type of bond ionic metallic covalent
Brief Explanations
- Ionic Bonds: Form between atoms with large electronegativity differences, typically metals (left side, groups 1-2) and nonmetals (right side, groups 16-17) of the periodic table.
- Metallic Bonds: Form exclusively between metal atoms, which are located on the left and middle sections (groups 1-12, plus some post-transition metals) of the periodic table.
- Covalent Bonds: Form between atoms with small electronegativity differences, typically nonmetals (right side, groups 14-17) and sometimes between nonmetals and metalloids, or atoms of the same nonmetal element.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Ionic: Atoms from the metal region (left/groups 1-2) and nonmetal region (right/groups 16-17) of the periodic table.
- Metallic: Atoms from the metal region (left/middle of the periodic table, groups 1-12 + post-transition metals).
- Covalent: Atoms from the nonmetal region (right/groups 14-17) of the periodic table (can be same or different nonmetals, or nonmetals with metalloids).