QUESTION IMAGE
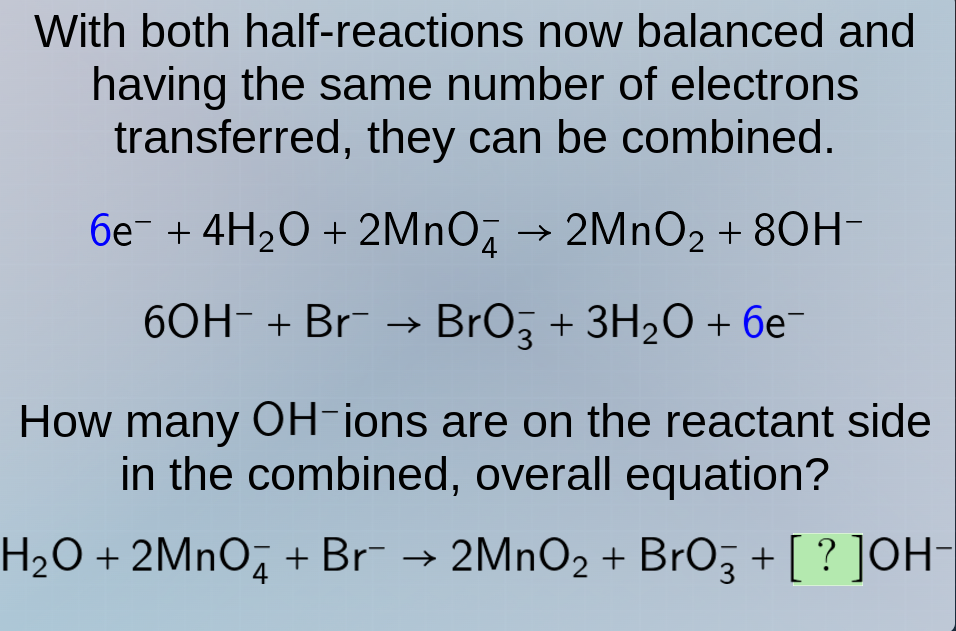
Question
with both half - reactions now balanced and having the same number of electrons transferred, they can be combined.
$6e^- + 4h_2o + 2mno_4^- \
ightarrow 2mno_2 + 8oh^-$
$6oh^- + br^- \
ightarrow bro_3^- + 3h_2o + 6e^-$
how many $oh^-$ ions are on the reactant side in the combined, overall equation?
$h_2o + 2mno_4^- + br^- \
ightarrow 2mno_2 + bro_3^- + ?oh^-$
\times2=6\) H (from \(3H_2O\)). So H is balanced in second half - reaction.
Now, when we add the two half - reactions:
Left side H: 8 (from first) + 6 (from second) = 14
Right side H: 8 (from first) + 6 (from second) = 14. So H is balanced.
Now, let's write the combined reaction:
\(6e^-+4H_2O + 2MnO_4^-+6OH^-+Br^-
ightarrow2MnO_2 + 8OH^-+BrO_3^-+3H_2O + 6e^-\)
Cancel \(6e^-\):
\(4
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\times2=6\) H (from \(3H_2O\)). So H is balanced in second half - reaction.
Now, when we add the two half - reactions:
Left side H: 8 (from first) + 6 (from second) = 14
Right side H: 8 (from first) + 6 (from second) = 14. So H is balanced.
Now, let's write the combined reaction:
\(6e^-+4H_2O + 2MnO_4^-+6OH^-+Br^-
ightarrow2MnO_2 + 8OH^-+BrO_3^-+3H_2O + 6e^-\)
Cancel \(6e^-\):
\(4