QUESTION IMAGE
Question
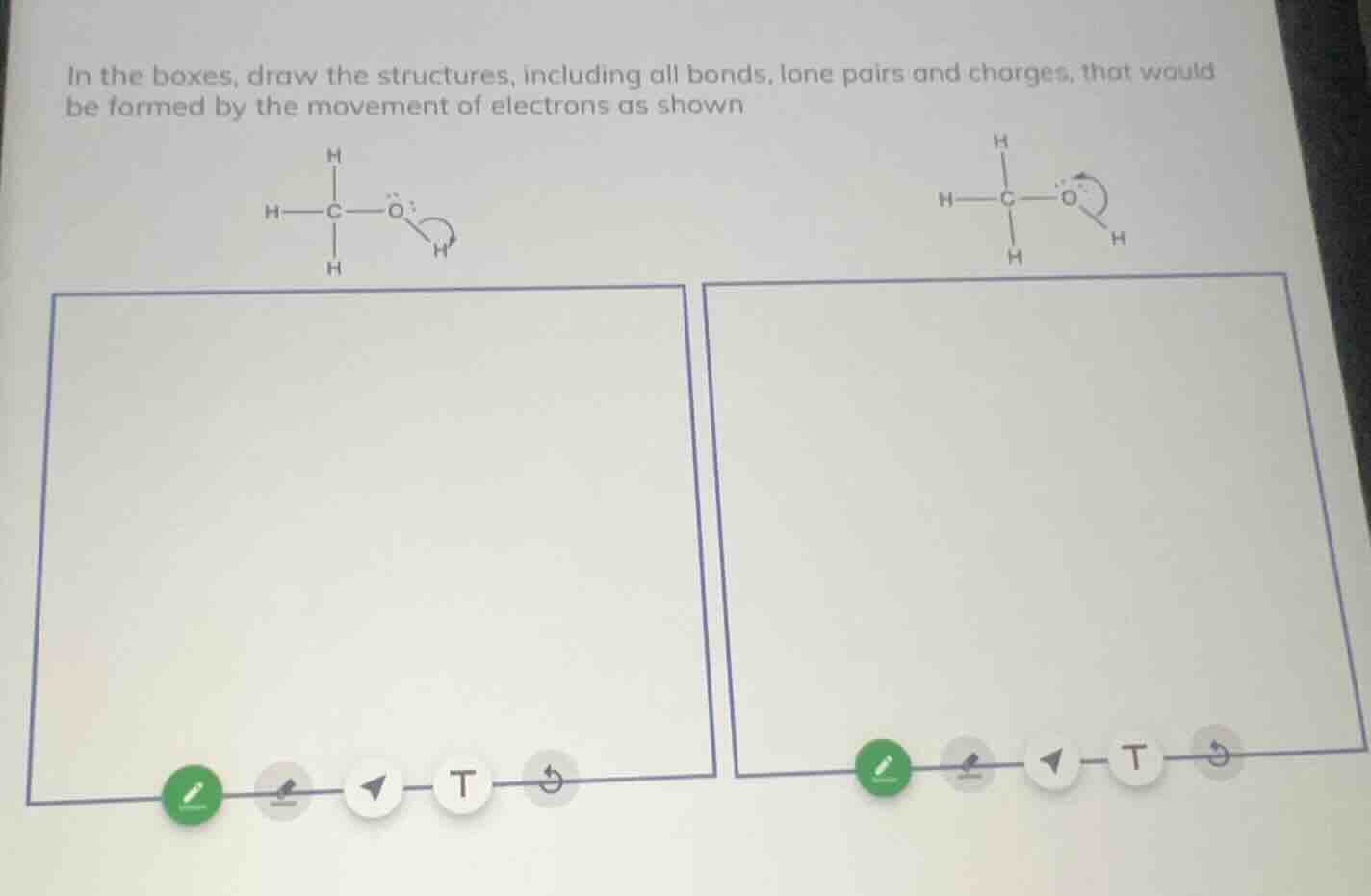
in the boxes, draw the structures, including all bonds, lone pairs and charges, that would be formed by the movement of electrons as shown
Brief Explanations
- For the first structure (left): The curved arrow shows the O-H bond's electrons moving to the oxygen atom. This breaks the O-H bond, leaving the oxygen with three lone pairs and a negative charge, and the hydrogen as a positively charged proton ($\text{H}^+$).
- For the second structure (right): The curved arrow shows a lone pair on oxygen forming a new bond to the adjacent carbon. This gives the oxygen three bonds (a positive charge, since oxygen's valence is 2) and the carbon retains its four bonds, with oxygen now having one lone pair.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Left box structure:
H
|
H - C - O:⁻
|
H(Plus a separate $\text{H}^+$; oxygen has 3 lone pairs, negative charge)
Right box structure:
H
|
H - C⁺ - O - H
|
H(Oxygen has 1 lone pair, positive charge; carbon has four bonds, no charge)