QUESTION IMAGE
Question
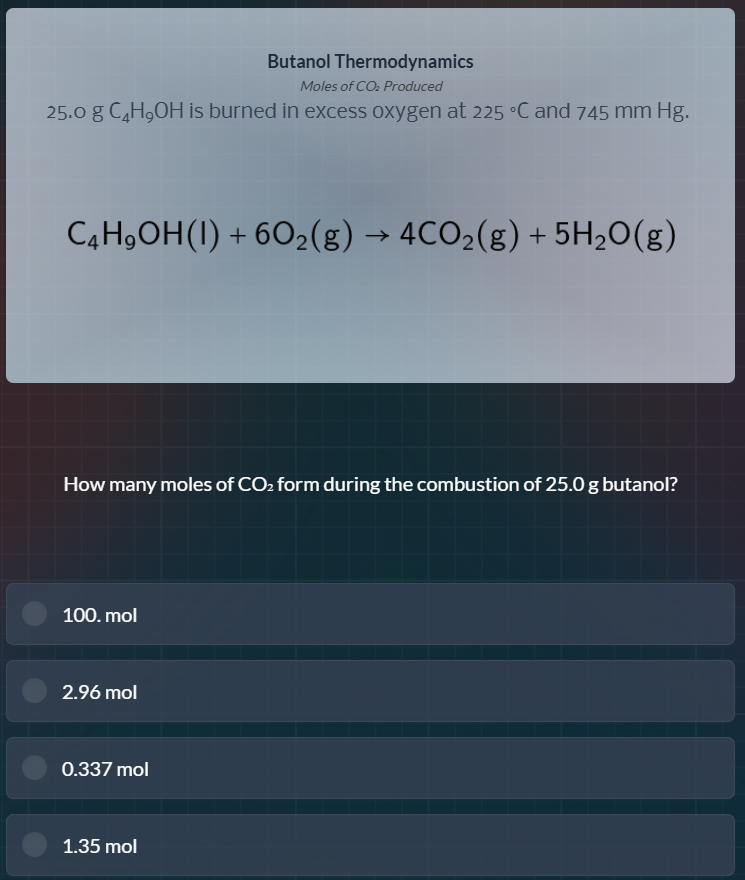
butanol thermodynamics
moles of co₂ produced
25.0 g c₄h₉oh is burned in excess oxygen at 225 °c and 745 mm hg.
c₄h₉oh(l) + 6o₂(g) → 4co₂(g) + 5h₂o(g)
how many moles of co₂ form during the combustion of 25.0 g butanol?
- mol
2.96 mol
0.337 mol
1.35 mol
Step1: Calculate molar mass of \( C_4H_9OH \)
Molar mass of \( C \): \( 12.01 \, \text{g/mol} \), \( H \): \( 1.008 \, \text{g/mol} \), \( O \): \( 16.00 \, \text{g/mol} \)
Molar mass \( = 4\times12.01 + 10\times1.008 + 16.00 = 74.12 \, \text{g/mol} \)
\( \text{Moles of } C_4H_9OH = \frac{25.0 \, \text{g}}{74.12 \, \text{g/mol}} \approx 0.337 \, \text{mol} \)
Step2: Use stoichiometry from reaction
Reaction: \( C_4H_9OH + 6O_2
ightarrow 4CO_2 + 5H_2O \)
Mole ratio \( C_4H_9OH : CO_2 = 1 : 4 \)
\( \text{Moles of } CO_2 = 0.337 \, \text{mol} \times 4 = 1.35 \, \text{mol} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.35 mol