QUESTION IMAGE
Question
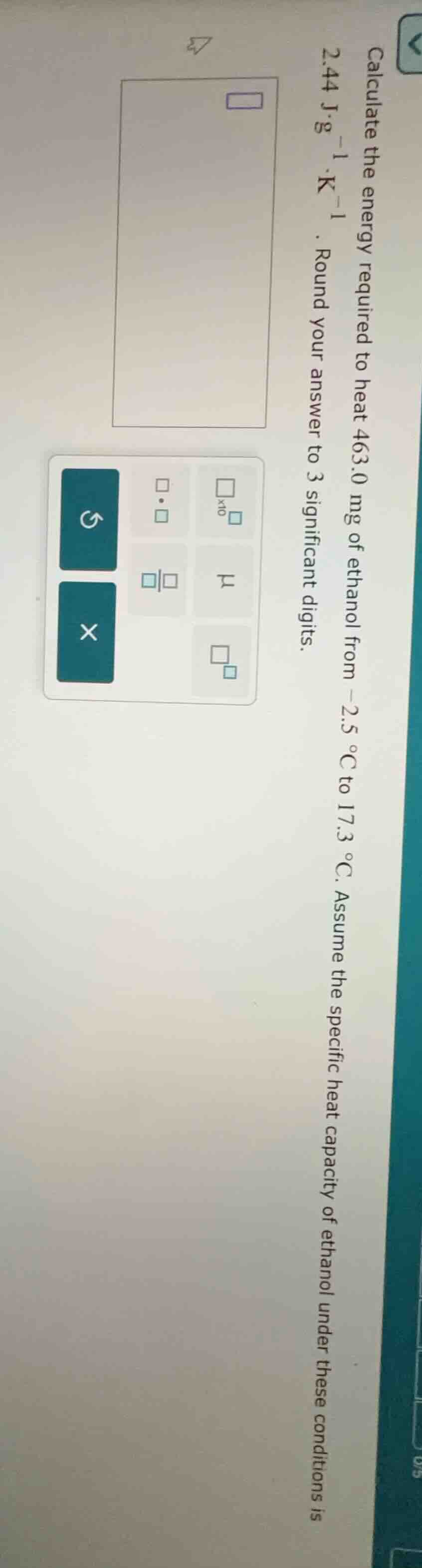
calculate the energy required to heat 463.0 mg of ethanol from -2.5 °c to 17.3 °c. assume the specific heat capacity of ethanol under these conditions is 2.44 j·g⁻¹·k⁻¹. round your answer to 3 significant digits.
Step1: Convert mass to grams
The mass of ethanol is \( 463.0 \, \text{mg} \). Since \( 1 \, \text{g} = 1000 \, \text{mg} \), we convert milligrams to grams:
\( m = 463.0 \, \text{mg} \times \frac{1 \, \text{g}}{1000 \, \text{mg}} = 0.4630 \, \text{g} \)
Step2: Calculate temperature change
The initial temperature \( T_i = -2.5 \, ^\circ\text{C} \) and final temperature \( T_f = 17.3 \, ^\circ\text{C} \). The temperature change \( \Delta T \) is:
\( \Delta T = T_f - T_i = 17.3 \, ^\circ\text{C} - (-2.5 \, ^\circ\text{C}) = 19.8 \, ^\circ\text{C} \) (or \( 19.8 \, \text{K} \), since the change in Celsius equals the change in Kelvin).
Step3: Use the heat formula \( q = mc\Delta T \)
The specific heat \( c = 2.44 \, \text{J·g}^{-1}·\text{K}^{-1} \), mass \( m = 0.4630 \, \text{g} \), and \( \Delta T = 19.8 \, \text{K} \). Substitute into the formula:
\( q = (0.4630 \, \text{g}) \times (2.44 \, \text{J·g}^{-1}·\text{K}^{-1}) \times (19.8 \, \text{K}) \)
First, multiply \( 0.4630 \times 2.44 \approx 1.13 \) (rounded for intermediate step). Then, \( 1.13 \times 19.8 \approx 22.4 \) (more precisely, \( 0.4630 \times 2.44 = 1.13072 \); \( 1.13072 \times 19.8 = 22.388256 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The energy required is approximately \( \boldsymbol{22.4 \, \text{J}} \) (rounded to 3 significant figures).