QUESTION IMAGE
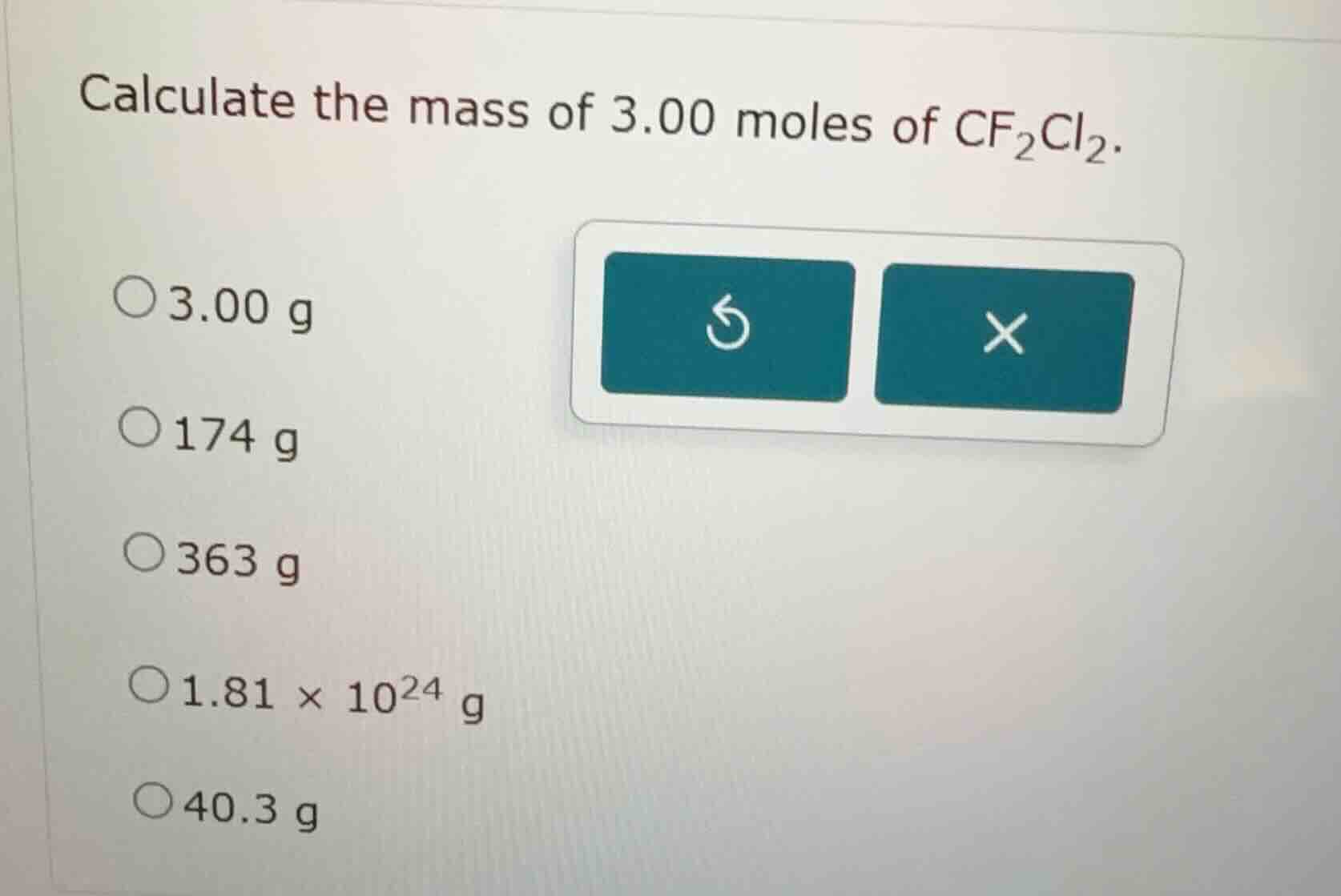
Question
calculate the mass of 3.00 moles of cf₂cl₂.
○3.00 g
○174 g
○363 g
○1.81 × 10²⁴ g
○40.3 g
Step1: Calculate molar mass of \( CF_2Cl_2 \)
Molar mass of \( C \): \( 12.01 \, g/mol \), \( F \): \( 19.00 \, g/mol \) (2 atoms), \( Cl \): \( 35.45 \, g/mol \) (2 atoms).
Molar mass \( = 12.01 + 2\times19.00 + 2\times35.45 = 12.01 + 38.00 + 70.90 = 120.91 \, g/mol \).
Step2: Use \( \text{Mass} = \text{Moles} \times \text{Molar Mass} \)
Given moles \( = 3.00 \, mol \), molar mass \( = 120.91 \, g/mol \).
Mass \( = 3.00 \, mol \times 120.91 \, g/mol = 362.73 \, g \approx 363 \, g \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
363 g (corresponding to the option "363 g")