QUESTION IMAGE
Question
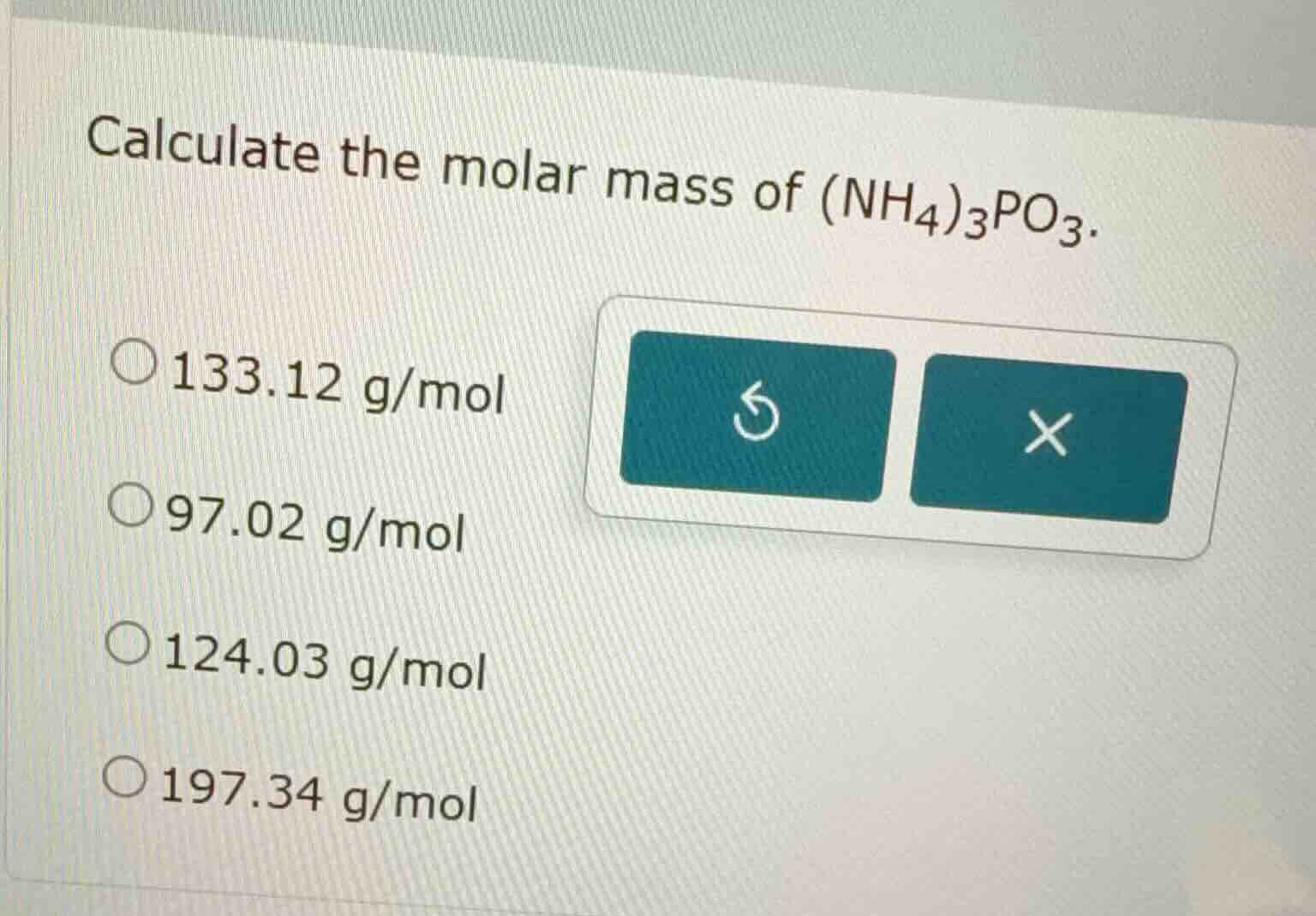
calculate the molar mass of (nh₄)₃po₃. 133.12 g/mol 97.02 g/mol 124.03 g/mol 197.34 g/mol
Step1: Identify atomic masses
Atomic masses (from periodic table): N = 14.01 g/mol, H = 1.008 g/mol, P = 30.97 g/mol, O = 16.00 g/mol.
Step2: Calculate moles of each atom
In \((NH_4)_3PO_3\):
- N: \(3\) (from \((NH_4)_3\))
- H: \(4 \times 3 = 12\) (4 per \(NH_4\), 3 groups)
- P: \(1\)
- O: \(3\)
Step3: Multiply by atomic masses
- N: \(3 \times 14.01 = 42.03\)
- H: \(12 \times 1.008 = 12.096\)
- P: \(1 \times 30.97 = 30.97\)
- O: \(3 \times 16.00 = 48.00\)
Step4: Sum the masses
Total molar mass = \(42.03 + 12.096 + 30.97 + 48.00\)
= \(42.03 + 12.096 = 54.126\); \(54.126 + 30.97 = 85.096\); \(85.096 + 48.00 = 133.096 \approx 133.12\) g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
133.12 g/mol