QUESTION IMAGE
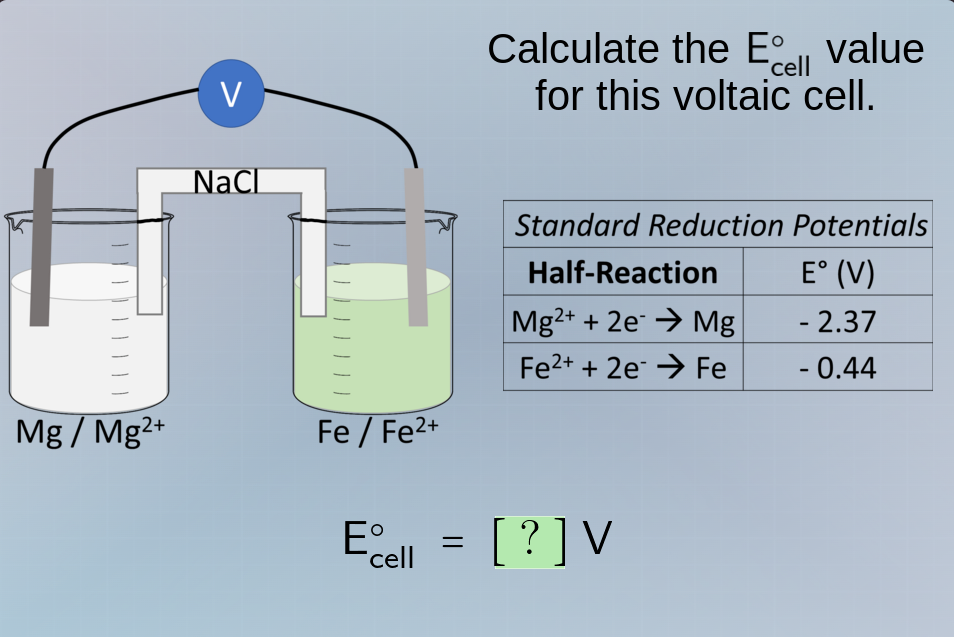
Question
calculate the $\ce{e^{\circ}_{cell}}$ value for this voltaic cell.
standard reduction potentials
| half-reaction | $\ce{e^{\circ}}$ (v) |
|---|
| $\ce{mg^{2+} + 2e^- \
ightarrow mg}$ | - 2.37 |
| $\ce{fe^{2+} + 2e^- \
ightarrow fe}$ | - 0.44 |
$\ce{e^{\circ}_{cell}} = ?$ v
Step1: Identify Anode and Cathode
In a voltaic cell, the more negative reduction potential indicates the stronger reducing agent (undergoes oxidation, anode). Mg has a more negative \( E^\circ \) (\( -2.37 \, V \)) than Fe (\( -0.44 \, V \)), so Mg is oxidized (anode: \( \text{Mg}
ightarrow \text{Mg}^{2+} + 2e^- \), reverse of its reduction half - reaction), and Fe\(^{2+}\) is reduced (cathode: \( \text{Fe}^{2+}+ 2e^-
ightarrow \text{Fe} \), same as its reduction half - reaction).
Step2: Calculate \( E^\circ_{\text{cell}} \)
The formula for the standard cell potential is \( E^\circ_{\text{cell}}=E^\circ_{\text{cathode (reduction)}} - E^\circ_{\text{anode (reduction)}} \). Here, \( E^\circ_{\text{cathode (reduction)}}=- 0.44 \, V \) (for \( \text{Fe}^{2+}+2e^-
ightarrow \text{Fe} \)) and \( E^\circ_{\text{anode (reduction)}}=-2.37 \, V \) (for \( \text{Mg}^{2+}+2e^-
ightarrow \text{Mg} \)).
Substitute the values into the formula:
\( E^\circ_{\text{cell}}=- 0.44-(-2.37) \)
\( E^\circ_{\text{cell}}=-0.44 + 2.37 \)
\( E^\circ_{\text{cell}} = 1.93 \, V \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 1.93 \)