QUESTION IMAGE
Question
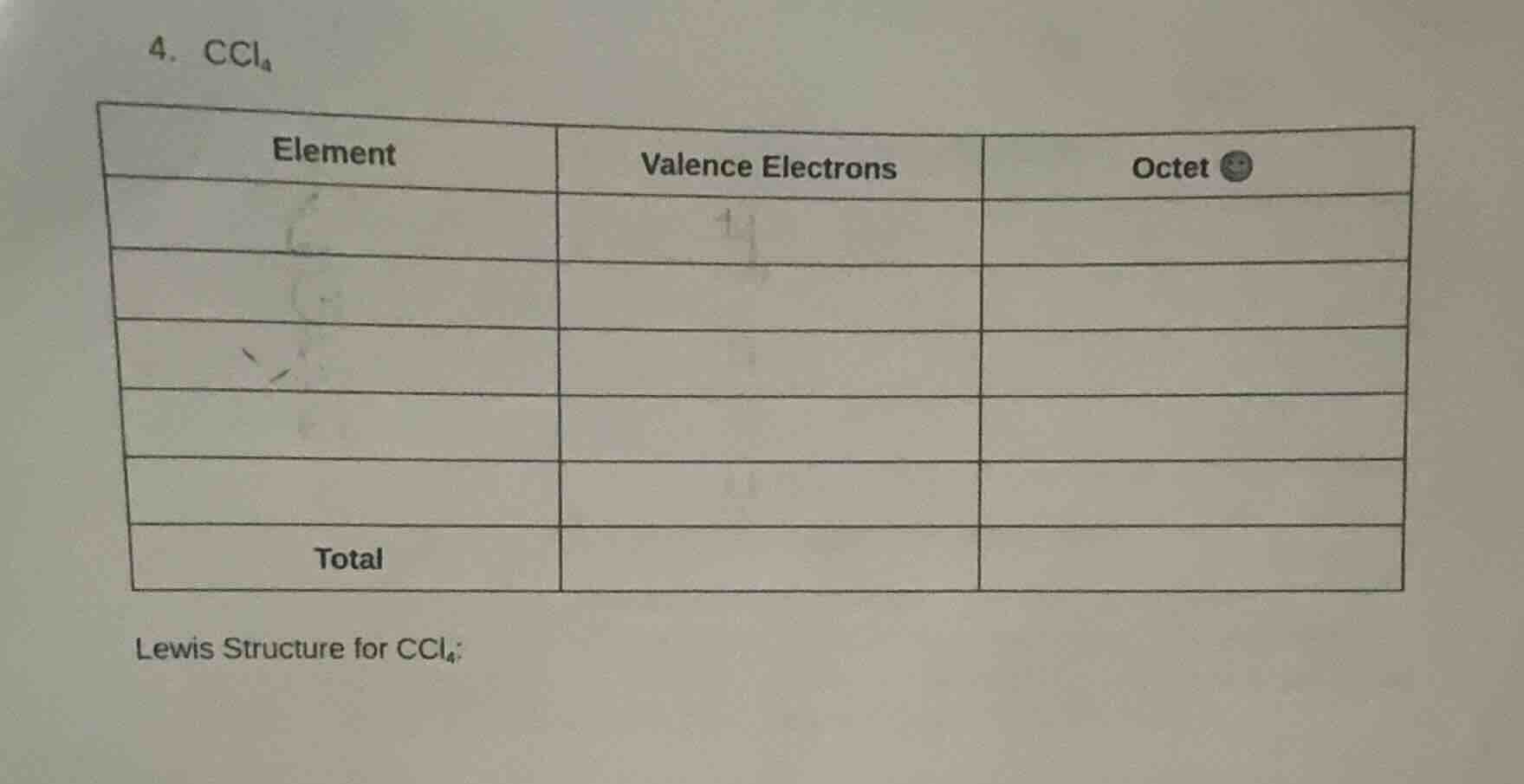
- ccl₄
| element | valence electrons | octet |
|---|---|---|
| total |
lewis structure for ccl₄:
Step1: Identify elements in \( \text{CCl}_4 \)
\( \text{CCl}_4 \) has 1 Carbon (C) and 4 Chlorine (Cl) atoms.
Step2: Valence electrons of C
Carbon is in group 14, so valence electrons \( = 4 \). Octet: Carbon needs 4 more electrons to complete octet (already has 4, so needs 4).
Step3: Valence electrons of each Cl
Chlorine is in group 17, valence electrons \( = 7 \). Octet: Each Cl needs 1 more electron to complete octet (has 7, needs 1).
Step4: Fill table for each element
- Carbon (C): Valence Electrons = 4, Octet (needs) = 4
- Chlorine (Cl) - 1st: Valence Electrons = 7, Octet (needs) = 1
- Chlorine (Cl) - 2nd: Valence Electrons = 7, Octet (needs) = 1
- Chlorine (Cl) - 3rd: Valence Electrons = 7, Octet (needs) = 1
- Chlorine (Cl) - 4th: Valence Electrons = 7, Octet (needs) = 1
Step5: Calculate total valence electrons
Total valence electrons \( = 4 + (4 \times 7) = 4 + 28 = 32 \).
Total octet (needs) \( = 4 + (4 \times 1) = 4 + 4 = 8 \) (since C needs 4 and each Cl needs 1, 4 Cls need 4).
Step6: Lewis Structure of \( \text{CCl}_4 \)
Carbon is central atom. It forms single bonds with 4 Cl atoms. Each Cl has 3 lone pairs (6 electrons) and 1 bonding pair (2 electrons), so each Cl has octet (8 electrons: 6 lone + 2 bonding). Carbon has 4 bonding pairs (8 electrons), so octet is complete. The Lewis structure is \( \text{Cl} - \text{C} - \text{Cl} \) (with Cl atoms at each corner, each Cl having 3 lone pairs, and C in the center with 4 single bonds to Cls).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Table Filling:
| Element | Valence Electrons | Octet (Needs) |
|---|---|---|
| Cl | 7 | 1 |
| Cl | 7 | 1 |
| Cl | 7 | 1 |
| Cl | 7 | 1 |
| Total | \( 4 + 4 \times 7 = 32 \) | \( 4 + 4 \times 1 = 8 \) |
Lewis Structure for \( \text{CCl}_4 \):
Carbon (C) is at the center, bonded to four Chlorine (Cl) atoms via single bonds. Each Cl atom has three lone pairs of electrons (6 electrons) and one bonding pair (2 electrons) with C, so each Cl has an octet (8 electrons). Carbon has four bonding pairs (8 electrons), completing its octet. The structure can be represented as:
\( \text{Cl}:\!\!\!\!\overset{\cdot \cdot}{\underset{\cdot \cdot}{{\text{Cl}}}} - \text{C} - \overset{\cdot \cdot}{\underset{\cdot \cdot}{{\text{Cl}}}}:\!\!\!\!\text{Cl} \) (with each Cl having 3 lone pairs, and C in the middle with 4 single bonds to Cls).